The role of UVR and vitamin D on T cells and inflammatory bowel disease
Abstract
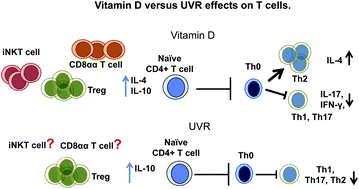
Vitamin D deficiency is associated with the development of inflammatory bowel disease (IBD). In experimental IBD the targets of vitamin D that result in protection from IBD include gut epithelial cells, innate immune cells, T cells, and the microbiota. Ultraviolet radiation (UVR) induces production of vitamin D in the skin and suppresses T cell responses in the host. There is limited data demonstrating an effect of UVR on experimental IBD but the mechanisms of UVR suppression in IBD have not been defined. There are several shared effects of vitamin D and UVR on T cells including inhibition of proliferation and suppression of IFN-γ and IL-17 producing T cells. Conversely UVR decreases and vitamin D increases IL-4 production from T cells. Together the data suggest that UVR suppression of T cells and potentially IBD are both vitamin D dependent and independent.
- This article is part of the themed collections: 2017 Perspective articles collection and The health benefits of UV radiation exposure through vitamin D production or non-vitamin D pathways

 Please wait while we load your content...
Please wait while we load your content...