Pressure-driven opening of carbon nanotubes
Abstract
The closing and opening of carbon nanotubes (CNTs) is essential for their applications in nanoscale chemistry and biology. We report reactive molecular dynamics simulations of CNT opening triggered by internal pressure of encapsulated gas molecules. Confined argon generates 4000 bars of pressure inside capped CNT and lowers the opening temperature by 200 K. Chemical interactions greatly enhance the efficiency of CNT opening: fluorine-filled CNTs open by fluorination of carbon bonds at temperature and pressure that are 700 K and 1000 bar lower than for argon-filled CNTs. Moreover, pressure induced CNT opening by confined gases leaves the CNT cylinders intact and removes only the fullerene caps, while the empty CNT decomposes completely. In practice, the increase in pressure can be achieved by near-infrared light, which penetrates through water and biological tissues and is absorbed by CNTs, resulting in rapid local heating. Spanning over a thousand of bars and Kelvin, the reactive and non-reactive scenarios of CNT opening represent extreme cases and allow for a broad experimental control over properties of the CNT interior and release conditions of the confined species. The detailed insights into the thermodynamic conditions and chemical mechanisms of the pressure-induced CNT opening provide practical guidelines for the development of novel nanoreactors, catalysts, photo-catalysts, imaging labels and drug delivery vehicles.
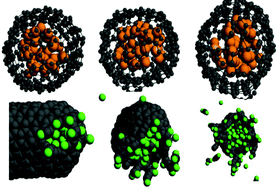
- This article is part of the themed collection: 2016 Nanoscale HOT Article Collection

 Please wait while we load your content...
Please wait while we load your content...