Determination of the binding constant of specific interactions and binding target concentration simultaneously using a general chemiluminescence method†
Abstract
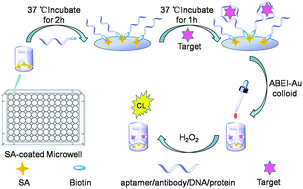
The measurement of the binding constant of specific interactions and concentration of a target is of considerable importance in clinical diagnosis, therapy, bioassays and drug design. The development of methods that combine high sensitivity with generalization and simplicity for the measurement of both binding constant and target concentration is highly desirable. Previously, we developed a label-free chemiluminescence (CL) strategy for the measurement of the target concentration and binding constants between DNA aptamers and target simultaneously based on the fact that the target could enhance the CL produced by the reaction of the N-(4-aminobutyl)-N-ethylisoluminol (ABEI) functionalized gold colloid with H2O2. In this study, the enhancement and inhibition effect of various targets on the CL reaction is studied. The generalization of the proposed CL strategy for various targets is also explored. The results demonstrate that the proposed CL strategy is suitable for targets that can cause a change in CL intensity, which includes enhancement and inhibition. It could be applied for the measurement of the dissociation constants of aptamer-binding targets, antibody–antigen complexes, protein-binding small molecules and double-strand DNA hybrids from the millimole to picomole level. It could also be used for the sensitive determination of target concentration, including 2,4,6-trinitrotoluene (TNT), dopamine, tetracycline, human IgG (hIgG), tuberculosis (TB) DNA and mannose, with the detection limit of 0.93 nM–4.1 fM. This strategy is of great potential in fundamental research as well as in applications in life sciences.

 Please wait while we load your content...
Please wait while we load your content...