CdS nanorod–MFe2O4 (M = Zn, Co and Ni) nanocomposites: a heterojunction synthesis strategy to mitigate environmental deterioration
Abstract
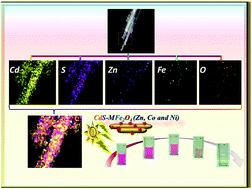
A facile strategy to encrust MFe2O4 (M = Zn, Co and Ni) nanoparticles over CdS nanorods via a two-step solvothermal method has been reported. The ferrite–CdS nanocomposites (NCs) were characterized using powder X-ray diffraction (XRD) and Fourier transform infrared (FT-IR) spectroscopy. A shift in the peak corresponding to the (311) plane confirmed the presence of different metal ions in the spinel ferrite lattice. However, no variation in the peak of CdS was observed which indicates that the phase and morphology of the CdS nanorods remain unaltered after hydrothermal treatment. High resolution transmission electron microscopy (HR-TEM) analysis revealed the efficacious attachment of the nanoferrite on the CdS nanorods. Optical studies of the NC samples provided information about the fabrication of the visible light responsive photocatalyst and covered the solar spectrum from 525 nm to 737 nm. From magnetic studies, CdS–CoFe2O4 and CdS–NiFe2O4 were found to be ferromagnetic in nature with a saturation magnetization of 26.4 and 15.5 emu g−1, respectively. Interestingly, a transition from ferromagnetic to super-paramagnetic was observed for the ZnFe2O4 loaded CdS nanorods. The photo-catalytic activities of the nanocomposites were studied by carrying out photo-degradation of rhodamine B and methylene blue dyes under visible-light irradiation. The maximum activity was observed in the case of the CdS–ZnFe2O4 nanocomposites.

 Please wait while we load your content...
Please wait while we load your content...