Direct synthesis of V–W–Ti nanoparticle catalysts for selective catalytic reduction of NO with NH3
Abstract
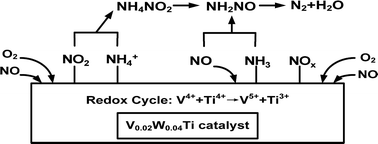
A series of V–W–Ti nanoparticle catalysts with variable V doping amounts were directly synthesized by the sol–gel method, and their catalytic performances were tested for the selective catalytic reduction of NO with ammonia. The catalysts were characterized by means of XRD, Raman, BET, TEM, SEM, NH3-TPD, H2-TPR and XPS. SCR kinetic studies were conducted to understand the mechanistic features of V–W–Ti catalysts. It was found that the V0.02W0.04Ti catalyst exhibited the highest NO conversion and the lowest apparent activation energy. The characterization results showed that V was incorporated into the TiO2 framework and the redox cycle of V4+ + Ti4+ → V5+ + Ti3+ existed over the V–W–Ti catalysts. A high concentration of reducible and distorted V species could account for the excellent NH3-SCR catalytic performance of the V0.02W0.04Ti catalyst. In situ FT-IR spectroscopy was performed to investigate the mechanism of the NH3-SCR reaction over the V0.02W0.04Ti catalyst. Experimental results showed that both Lewis and Brønsted acid sites over the V0.02W0.04Ti catalyst were involved in the NH3-SCR reaction. The adsorption of nitrate species was significantly limited and the adsorbed NO2 gaseous molecules were easily formed over the V0.02W0.04Ti catalyst, which resulted in the high catalytic activity at low temperature.

 Please wait while we load your content...
Please wait while we load your content...