Phosphorelay of non-orthodox two component systems functions through a bi-molecular mechanism in vivo: the case of ArcB†
Abstract
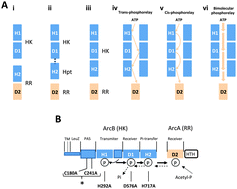
Two-component systems play a central part in bacterial signal transduction. Phosphorelay mechanisms have been linked to more robust and ultra-sensitive signalling dynamics. The molecular machinery that facilitates such a signalling is, however, only understood in outline. In particular the functional relevance of the dimerization of a non-orthodox or hybrid histidine kinase along which the phosphorelay takes place has been a subject of debate. We use a combination of molecular and genetic approaches, coupled to mathematical and statistical modelling, to demonstrate that the different possible intra- and inter-molecular mechanisms of phosphotransfer are formally non-identifiable in Escherichia coli expressing the ArcB non-orthodox histidine kinase used in anoxic redox control. In order to resolve this issue we further analyse the mathematical model in order to identify discriminatory experiments, which are then performed to address cis- and trans-phosphorelay mechanisms. The results suggest that exclusive cis- and trans-mechanisms will not be operating, instead the functional phosphorelay is likely to build around a sequence of allosteric interactions among the domain pairs in the histidine kinase. This is the first detailed mechanistic analysis of the molecular processes involved in non-orthodox two-component signalling and our results suggest strongly that dimerization facilitates more discriminatory proof-reading of external signals, via these allosteric reactions, prior to them being further processed.

 Please wait while we load your content...
Please wait while we load your content...