Open Access Article
Open Access ArticleCreative Commons Attribution 3.0 Unported Licence
Ultrafine and respirable particle exposure during vehicle fire suppression
Douglas E.
Evans
*a and
Kenneth W.
Fent
b
aChemical Exposure and Monitoring Branch, Division of Applied Research and Technology, National Institute for Occupational Safety and Health, Centers for Disease Control and Prevention, 1090 Tusculum Avenue, MS R-7, Cincinnati, Ohio, USA. E-mail: dje3@cdc.gov; Fax: +1 513 841 4545; Tel: +1 513 841 4407
bHazard Evaluations and Technical Assistance Branch, Division of Surveillance, Hazard Evaluations and Field Studies, National Institute for Occupational Safety and Health, Centers for Disease Control and Prevention, Cincinnati, Ohio, USA
First published on 26th August 2015
Abstract
Vehicle fires are a common occurrence, yet few studies have reported exposures associated with burning vehicles. This article presents an assessment of firefighters' potential for ultrafine and respirable particle exposure during vehicle fire suppression training. Fires were initiated within the engine compartment and passenger cabins of three salvaged vehicles, with subsequent water suppression by fire crews. Firefighter exposures were monitored with an array of direct reading particle and air quality instruments. A flexible metallic duct and blower drew contaminants to the instrument array, positioned at a safe distance from the burning vehicles, with the duct inlet positioned at the nozzle operator's shoulder. The instruments measured the particle number, active surface area, respirable particle mass, photoelectric response, aerodynamic particle size distributions, and air quality parameters. Although vehicle fires were suppressed quickly (<10 minutes), firefighters may be exposed to short duration, high particle concentration episodes during fire suppression, which are orders of magnitude greater than the ambient background concentration. A maximum transient particle concentration of 1.21 × 107 particles per cm3, 170 mg m−3 respirable particle mass, 4700 μm2 cm−3 active surface area and 1400 (arbitrary units) in photoelectric response were attained throughout the series of six fires. Expressed as fifteen minute time-weighted averages, engine compartment fires averaged 5.4 × 104 particles per cm3, 0.36 mg m−3 respirable particle mass, 92 μm2 cm−3 active particle surface area and 29 (arbitrary units) in photoelectric response. Similarly, passenger cabin fires averaged 2.04 × 105 particles per cm3, 2.7 mg m−3 respirable particle mass, 320 μm2 cm−3 active particle surface area, and 34 (arbitrary units) in photoelectric response. Passenger cabin fires were a greater potential source of exposure than engine compartment fires. The wind direction and the relative position of the fire crew to the stationary burning vehicle played a primary role in fire crews' potential for exposure. We recommend that firefighters wear self-contained breathing apparatus during all phases of the vehicle fire response to significantly reduce their potential for particulate, vapor, and gaseous exposures.
Environmental impactVehicle fires are typically intense, short duration fires that have the potential to release a plethora of combustion and thermally degraded products into the environment; the atmosphere being most notably and visibly affected. Vehicle fire residues and contaminated water from fire suppression may also enter into aquatic and soil systems. Uncontrolled combustion and thermal degradation of plastics, composites, upholstery, alloys, paints, coatings, sealants, adhesives, tires, belts, wires, batteries, lubricants, coolants, and fuels can result in the release of a mixture of toxic compounds. First responders to a vehicle fire are potentially exposed to these compounds. Air quality is also adversely affected, in the locale or downwind of a vehicle fire. |
Introduction
From 2003 to 2007, an average of 300![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 highway vehicle fires occurred annually in the United States, representing 17% of all fire department responses.1 Of 300
000 highway vehicle fires occurred annually in the United States, representing 17% of all fire department responses.1 Of 300![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 highway vehicle fires, 86% involved passenger vehicles.2 Despite their regular frequency, relatively few studies have been conducted for vehicle fires, and those reported, have focused on emissions and combustion processes rather than potential hazardous exposures to firefighters.3–6
000 highway vehicle fires, 86% involved passenger vehicles.2 Despite their regular frequency, relatively few studies have been conducted for vehicle fires, and those reported, have focused on emissions and combustion processes rather than potential hazardous exposures to firefighters.3–6
Exposure to particulate matter as a component of ambient air pollution is widely recognized as a cause for adverse cardiovascular and respiratory health effects in the general population.7 Firefighters are frequently exposed to particulate matter within smoke as a consequence of their fire suppression duties. Particulate matter exposure may play a crucial role in firefighters acquiring two diseases: cancer and heart disease. Two recent studies have strengthened the association between firefighting, as an occupation, and an increase in the incidence of certain cancers found in the respiratory, digestive, and urinary systems when compared to the general population.8,9 The inhaled particulate matter during firefighting could be a causative factor for some of these cancers.
A second health outcome potentially related to particulate exposure is cardiac events. Each year in the United States, approximately 35 to 40 firefighters succumb to sudden cardiac death on duty, while more than 600 suffer non-fatal cardiac events.10,11 Of the approximately 1.15 million firefighters (350![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 career and 800
000 career and 800![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 volunteer) in the United States, there are approximately 100 firefighter fatalities in any given year and between one third10 to one half12 are due to a sudden cardiac event. Particulate exposure in combination with other cardiovascular stressors is believed to play a crucial role in some of these sudden cardiac events.13
000 volunteer) in the United States, there are approximately 100 firefighter fatalities in any given year and between one third10 to one half12 are due to a sudden cardiac event. Particulate exposure in combination with other cardiovascular stressors is believed to play a crucial role in some of these sudden cardiac events.13
The study described here was conducted as a NIOSH health hazard evaluation14 concerning firefighters' potential exposures during vehicle fire suppression training. Training exercises provide a realistic and unique opportunity to observe and monitor firefighters during multiple live fires. Although self-contained breathing apparatus (SCBA) can significantly or completely mitigate inhalation exposure when fitted and functioning, they are not always worn or may be doffed prematurely. Dermal absorption may also contribute to the systemic dose of some toxicants.15 This paper is the second of a two part series. This paper estimates a firefighters' potential exposure to ultrafine and respirable particles during vehicle fire suppression using direct reading instruments. The first paper reported potential chemical vapor and gas exposures for the same series of vehicle fires.16
Particle monitoring described here focused on the application of direct reading instruments, primarily because fire responses were anticipated to be of short duration and sequential monitoring of each suppression phase was of particular interest. Multiple particle metrics were desired in parallel because various characteristics may contribute to particle toxicity. For example, the particle mass alone may not fully account for adverse health effects from fine particle exposure.17
Methods
Population, site, vehicles and fires
Nineteen firefighters (mostly volunteer), including fire chief and assistant fire chief from a small municipal fire department in Southwest Ohio, participated in the evaluation.14 Salvaged vehicles were used in the training exercises, which took place on a former industrial parking lot, surfaced with concrete. Training consisted of three phases: (1) startup or ignition—when the fire was ignited, allowed to build in intensity, and fire fighters approached the burning vehicle, (2) knockdown—when the fire (flames) was suppressed with a single water jet, and (3) overhaul—when the firefighters searched for and suppressed flare-ups and remaining hot-spots. Engines and cabins of the vehicles were separately and sequentially ignited with signal flares and a small volume of gasoline (petroleum) as the accelerant. A single fire apparatus was present at the fire scene. The firefighters observed the fire from a safe distance for two to five minutes to let the fires build in intensity before suppression commenced. The duration for each suppression phase averaged thirty seconds for approach, as the firefighters positioned themselves at the burning vehicle, two minutes for knockdown, and two to three minutes for overhaul.Three firefighters were involved in the active suppression of the vehicle fires: a nozzle operator, a backup, and an officer. The nozzle operator directed a single water jet, the backup assisted with handling the hose and the officer directed and assisted where needed. In addition, a fourth firefighter assisted with the particle sampling described here, by holding the sampling duct inlet close to the shoulder of the nozzle operator to estimate particle concentrations in the firefighters' breathing zone. All firefighters wore full turnout gear and SCBA throughout all three phases. Engine compartments and passenger cabins were sequentially ignited for: a 1994 Ford Aerostar® minivan, a 1986 Toyota Corolla® sedan, and a 1986 Toyota Celica® coupe, totaling six fires. The belts, fluids, batteries, cushions, and upholstery were present in each vehicle, but fuel tanks had been previously drained.
Particle sampling
Particles were sampled in close proximity to the firefighters suppressing fires with the use of a 7.6 meter length of 15 cm diameter flexible aluminum ducting attached to a mobile particle sampling platform previously described in detail.18 The platform, custom configured for this application (Fig. 1), allowed simultaneous and continuous measurement of particles generated from fires in real time by multiple instruments and metrics. A downstream blower (Ramfan UB20, Euramco Safety, Spring Valley, California) drew combustion contaminants through the ducting to the instruments. A mean velocity of 6.0 m s−1 was attained within the flexible duct, resulting in a contaminant residence time of 1.3 s from the duct inlet to instrument sampling probes. The flow within the duct was turbulent (Reynolds number, Re ∼60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000). Stainless steel sampling probes with sharp beveled inlets and gentle radii were sized and oriented toward the duct inlet to minimize particle sampling artifacts. Short lengths of conductive silicone tubing were used to connect sampling probes to instruments. The flexible sampling duct can be seen in Fig. 2, being held by one of the fire crew during knockdown for the first vehicle engine fire.
000). Stainless steel sampling probes with sharp beveled inlets and gentle radii were sized and oriented toward the duct inlet to minimize particle sampling artifacts. Short lengths of conductive silicone tubing were used to connect sampling probes to instruments. The flexible sampling duct can be seen in Fig. 2, being held by one of the fire crew during knockdown for the first vehicle engine fire.
 | ||
| Fig. 1 The particle instrument sampling platform with the sample duct. Sample probes were positioned within the central region of the duct facing the induced flow. | ||
Particle concentration metrics included the number, active surface area, respirable mass, photoelectric response, and size distribution from 7 nm to 10 μm. The particle number was provided by using a condensation particle counter (CPC, TSI 3007, Shoreview, Minnesota) with an inline HEPA filter capsule based dilution device.18,19 Anticipated high particle number concentrations during fires, required CPC dilution. The endcap of the filter capsule was predrilled, such that the major flow through the capsule passed through the filtration media, and a minor flow bypassed the filtration media through the drilled endcap. This resulted in a dilution ratio of 118 to one, determined, by switching between diluted and undiluted background aerosol. Particle number concentration data were subsequently corrected by this ratio, for the duration the inline diluter was fitted. The active particle surface area, respirable particle mass, photoelectric response and particle size distributions were provided by using a diffusion charger (DC 2000CE, EcoChem Analytics, Murrieta, California), a photometer (TSI DustTrak 8520) with Dorr-Oliver cyclone inlet positioned within the duct, a photoelectric aerosol sensor (PAS 2000CE, EcoChem Analytics), and an Electrical Low Pressure Impactor (ELPI, Dekati, Tampere, Finland), respectively. Air quality metrics (temperature, relative humidity, CO, and CO2 concentrations) were also monitored from within the duct (TSI Q-Trak Plus 8554). A one second data logging interval was selected for the condensation particle counter, photometer, ELPI and Q-Trak. The diffusion charger and photoelectric aerosol sensor required a minimum ten second logging interval. Environmental variables (temperature, relative humidity, wind speed and direction) were obtained with an onsite mobile weather station previously described.14
Results
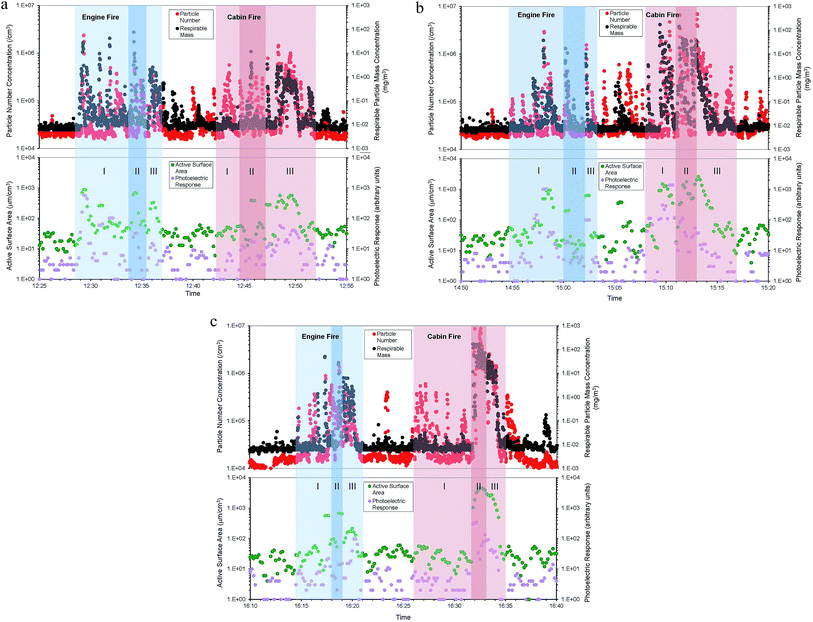
A light wind (4.2 km h−1) was present from a predominately southerly direction. Consequently, the firefighters approached the fires from either southeasterly (engine fires) or southerly (cabin fires) directions, which tended to take smoke away from the firefighters (some swirling or veering of the wind was also observed on occasions, as later discussed). In full sun, ambient air temperatures ranged from 24 to 26 °C, while the relative humidity remained relatively steady (mean average of 33%). A maximum relative humidity of ∼70% was measured within the duct following knockdown of the vehicle 3 passenger cabin fire. Using IR thermometry, passenger cabin fires were hotter than engine compartment fires, and some metallic vehicle parts remained at elevated temperatures (>93 °C) after knockdown. All but one fire exceeded 540 °C. However, no significant temperature elevations were observed within the particle sampling duct.
Fig. 3a–c represent simultaneous time series for the various particle metrics (on logarithmic scales) throughout the six vehicle fires. Intense, but short duration smoke episodes can be observed, elevating particle concentrations orders of magnitude above those of background and smoke episodes not associated with a particular phase of fire response. Fifteen-minute time weighted average (TWA) concentrations from each of the vehicle fires are presented in Table 1. A maximum transient particle concentration of 1.21 × 107 cm−3 for particle number, 170 mg m−3 for respirable mass, 4700 μm2 cm−3 for active surface area and 1400 (arbitrary units) for photoelectric response were obtained throughout the series of six fires. Fig. 4 presents particle number concentrations (on a logarithmic scale) measured by the ELPI and segregated into stages within the ultrafine (<100 nm), fine (100 nm > 1 μm) and coarse (1 μm > 10 μm) particle size ranges. Although particle number concentrations are dominated by the ultrafine particle size fraction, coarse and fine particles also contribute during intense smoke episodes. Fig. 5 presents four particle size distributions obtained with the ELPI, averaged over at least 30 seconds duration. An ambient background particle size distribution was obtained before all car fires took place (approx. 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 10 to 12
10 to 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 20). Diesel exhaust plumes from the upwind fire apparatus influenced measurements on an intermittent basis and were particularly evident between vehicle 1 and 2 fires (Fig. 4) due to the proximal position of the apparatus. One such transient concentration increase (approx. 15
20). Diesel exhaust plumes from the upwind fire apparatus influenced measurements on an intermittent basis and were particularly evident between vehicle 1 and 2 fires (Fig. 4) due to the proximal position of the apparatus. One such transient concentration increase (approx. 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 35) is further characterized in Fig. 5 and suggests a primary mode at approximately 40 nm. An engine fire plume distribution was obtained during the 2nd vehicle engine fire (approx. 15
35) is further characterized in Fig. 5 and suggests a primary mode at approximately 40 nm. An engine fire plume distribution was obtained during the 2nd vehicle engine fire (approx. 15![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 00), and a cabin fire plume distribution was obtained during the 3rd passenger cabin fire (approx. 16
00), and a cabin fire plume distribution was obtained during the 3rd passenger cabin fire (approx. 16![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) :
:![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 32). The vehicle fire plume distributions exhibit a somewhat similar size profile, but also some significant differences. The engine compartment fire plume manifests a primary mode in the ultrafine size range (approx. 30 nm), whereas the passenger cabin fire plume exhibits a primary mode in the fine size range (approx. 250 nm), suggesting a dominant accumulation mode. The passenger cabin fire plume is close to one order of magnitude greater in the ultrafine and two orders of magnitude greater in the fine and coarse particle size ranges in number concentration than the engine compartment fire plume.
32). The vehicle fire plume distributions exhibit a somewhat similar size profile, but also some significant differences. The engine compartment fire plume manifests a primary mode in the ultrafine size range (approx. 30 nm), whereas the passenger cabin fire plume exhibits a primary mode in the fine size range (approx. 250 nm), suggesting a dominant accumulation mode. The passenger cabin fire plume is close to one order of magnitude greater in the ultrafine and two orders of magnitude greater in the fine and coarse particle size ranges in number concentration than the engine compartment fire plume.
| Number (cm−3) × 103 | Respirable mass (mg m−3) | Active surface area (μm2 cm−3) | Photoelectric response | CO (ppm) | |||
|---|---|---|---|---|---|---|---|
| Vehicle 1 | Engine | Mean | 56 | 0.53 | 100 | 30 | 1.3 |
| Max. | 2360 | 76 | 880 | 580 | 7.2 | ||
| Cabin | Mean | 89 | 0.13 | 110 | 10 | 0.6 | |
| Max. | 1440 | 12 | 560 | 60 | 1.7 | ||
| Vehicle 2 | Engine | Mean | 54 | 0.22 | 97 | 50 | 0.4 |
| Max. | 2950 | 37 | 940 | 1000 | 1.5 | ||
| Cabin | Mean | 198 | 2.0 | 350 | 72 | 1.8 | |
| Max. | 6950 | 170 | 2600 | 1400 | 8.8 | ||
| Vehicle 3 | Engine | Mean | 53 | 0.33 | 80 | 8 | 0.4 |
| Max. | 1380 | 51 | 680 | 97 | 1.6 | ||
| Cabin | Mean | 324 | 5.9 | 490 | 21 | 7.0 | |
| Max. | 12![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 100 100 |
170 | 4700 | 340 | 62 | ||
| Background | Mean | 11 | 0.007 | 6 | 2 | 0.1 | |
| Max. | 15 | 0.011 | 9 | 5 | 0.2 | ||
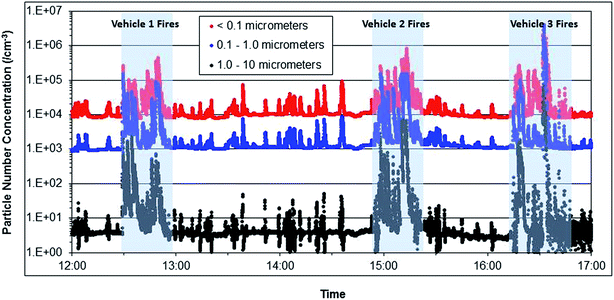 | ||
| Fig. 4 Size fractionated particle number as a time series for all fires, using the Electrical Low Pressure Impactor (ELPI). | ||
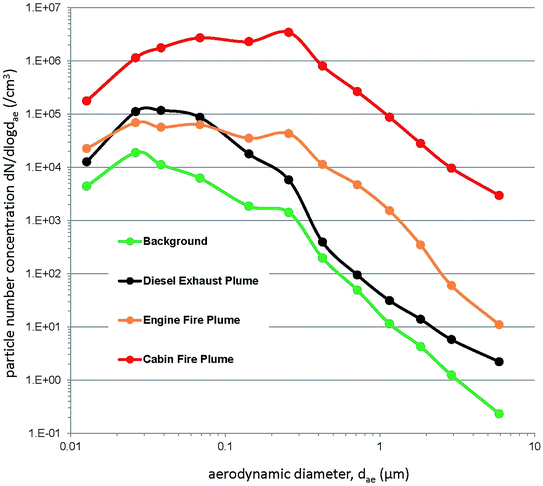 | ||
| Fig. 5 Particle size distributions acquired with the ELPI for background, diesel exhaust plume, engine compartment fire plume and passenger cabin fire plume. | ||
Discussion
Vehicle fires are common for the fire service. On average, one in seven fire department responses are to the 300![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000 annual vehicle fires.2 For those fire departments with major highways or road networks within their jurisdiction, the number of vehicle fire responses may be higher. Almost two thirds of all highway vehicle fires commence within the engine compartment, the running gear, or the wheel area. Furthermore, mechanical or electrical failures account for roughly three quarters of all fire causes.20
000 annual vehicle fires.2 For those fire departments with major highways or road networks within their jurisdiction, the number of vehicle fire responses may be higher. Almost two thirds of all highway vehicle fires commence within the engine compartment, the running gear, or the wheel area. Furthermore, mechanical or electrical failures account for roughly three quarters of all fire causes.20
Although some fire departments routinely employ SCBA during vehicle fire responses (similar to the fire crew in this study), this is not standard policy across all fire departments. When worn, fitted and functioning correctly, SCBA has an assigned protection factor of 1 × 104 and provides substantial protection, if not complete attenuation of toxicant inhalation exposures.21 Due to the anticipated short duration of the fire response, however, SCBA may not be donned by some crews and in volunteer fire departments, not all firefighters may possess SCBA. In a study of firefighters in Montreal QC, SCBA was used in only 6% of the time at all fires (including vehicle fires).22 More recently, and from a smaller study cohort, only approximately one third of firefighters from Chicago, IL reported SCBA use during vehicle fire knockdown.15 Restricted peripheral vision, fogging of the mask, hampered physical movement, weight of the SCBA ensemble, potential for heat stress and saving the compressed air tanks for longer duration structure fires are all factors that may influence the decision not to wear or earlier than optimal doffing of SCBA. Oftentimes, even if SCBA is initially donned for approach and knockdown, it may then be doffed for overhaul, leaving firefighters unprotected. It should also be noted that the SCBA can protect more than just the respiratory system. The face piece (mask) can also protect the eyes and face from radiant heat and airborne debris. During cabin fires in this study, windows shattered and tires exploded. Furthermore, if present within vehicles, air bags and other safety-related devices can deploy with the potential of flinging hot, sharp and/or toxic airborne debris out of the vehicle, potentially injuring unprotected fire crew members.
In this study, it was observed that the potential for particle exposure was primarily influenced by the position of the fire fighters relative to the burning vehicle and the light, southerly wind. Firefighters generally attacked the fire from an upwind or smoke clear position, as per standard fire department policies. However, this may not always be practical when access is restricted. During this study, the visible smoke plume would occasionally veer and engulf the fire crew. These veering plumes contributed to the highest concentration exposure events observed during the ignition/approach and knockdown phases, (Fig. 3a through c). The peak particle concentrations observed during the passenger cabin fire for vehicle 3 (Fig. 3c and Table 1) illustrate that exposures could persist into the overhaul phase, when a firefighter might remove SCBA. This particular plume event, which commenced during knockdown, lingered as the crew transitioned into overhaul. This plume was also largely responsible for driving the highest mean 15 minute TWA concentrations observed for the particle number, respirable particle mass and active surface area in this study (Table 1).
In addition, the fire crew would occasionally position themselves downwind of the water dowsed vehicle to enable better access to residual burning and smoldering components during overhaul. This was particularly evident following the knockdown of the passenger cabin fires, at which point the fire crew would be in the direct path of particulate concentrations that were falling but nevertheless elevated above those of the background (see Fig. 3a through c). The potential for elevated particle exposure was therefore observed during all phases of fire suppression, as evidenced by the increased particle number, respirable mass, and other metrics.
Although this article focuses on the particle exposure component, other exposures are likely, including exposures to volatile compounds. Fent and Evans,16 from the same series of vehicle fires as reported here, measured air concentrations of formaldehyde, acrolein, total reactive isocyanate groups, and CO near or above applicable ceiling limits or short-term exposure limits. According to exposure modelling for the most exposed firefighters, personal exposures to these volatile compounds were estimated to be nearly 10 times the acceptable levels on an additive effects basis.16 Bolstad-Johnson et al.23 examined firefighter exposures during overhaul activities in a series of residential structure fires in Phoenix, AZ. Several chemical exposure limits were exceeded, including ceiling limits for acrolein, CO, formaldehyde, and glutaraldehyde, and short term exposure limits for benzene, NO2, and SO2. In addition, PAH concentrations also exceeded exposure limits on an additive effects basis. Adverse respiratory effects have also been reported following structure fire overhaul.24
Respirable particles potentially serve as a vehicle to deposit adsorbed toxicants into the unciliated, gas exchange regions of the lung, where a large lung surface exists and clearance mechanisms are least effective. Respirable mass estimates from our study (Table 1), derived from direct-reading photometer measurements, averaged 0.36 mg m−3 for three engine fires and 2.7 mg m−3 for three cabin fires over 15 minute intervals. The background respirable mass concentration averaged 0.007 mg m−3. Higher particle mass concentrations obtained for passenger cabin fires can be explained by greater fuel loads, more extensive fires, and a longer suppression when compared to engine compartment fires. In comparison, Bolstad-Johnson et al.23 reported a mean average respirable dust concentration of 8.01 mg m−3 during the overhaul phase of residential structure fires. Fent et al.14 reported estimated PM10 (particulate with an aerodynamic diameter of less than 10 μm) mass concentration measurements averaging 340, 626, and 9.0 mg m−3 during active fires, knockdown, and overhaul, respectively, within burning structures. Our mean average results for the entire vehicle fire response are at significantly lower concentrations than those reported by either Bolstad-Johnson et al.23 during structural overhaul or those reported by Fent et al.15 during the sequential phases of the structural fire response. Combustion products confined within structures will generally enhance the magnitude of firefighters' exposure to toxicants. Particulate, gas, and vapor concentrations during fires have an inverse relationship with ventilation.25 In the open atmosphere, as is typical for the majority of vehicle fires, contaminants may be more quickly diluted, dispersed and/or moved away from the fire crew.
On a particle number concentration basis (Table 1), engine fires averaged 5.4 × 104 particles per cm3 and cabin fires averaged 2.04 × 105 particles per cm3. Background concentrations averaged 1.1 × 104 particles per cm3. Expressed as TWAs, passenger cabin fire particle number concentrations were always greater than engine compartment fire concentrations in our study. A greater fuel load consequently leads to a more extensive fire and a longer suppression, resulting in enhanced potential for particle number concentration exposure. Our 15 minute TWA results reported here are greater than the 1.96 × 104 particles per cm3 average reported by Baxter et al.6 during overhaul for a single vehicle cabin fire.
On a particle active surface area concentration basis, our results averaged 92 μm2 cm−3 overall for the engine fires and 320 μm2 cm−3 overall for the cabin fires. Similar to respirable particle mass and particle number, cabin fires were greater in the active particle surface area than engine fires. The active particle surface area generally follows the particle number more closely than the respirable particle mass18,26 and our results appear to be in keeping with these earlier observations. When displayed as time series (Fig. 3a through c), the active surface area generally follows particle number concentration despite differences in logging intervals for the diffusion charger and condensation particle counter, respectively. During carbon nanofiber manufacturing operations,18 active surface area concentrations averaged 640 μm2 cm−3; greater than the TWAs observed for engine or passenger cabin fire suppression in this study (Table 1). However, the maximum concentration of 4700 μm2 cm−3 attained during the vehicle 3 passenger cabin fire (Table 1) exceeded the maximum of 1440 μm2 cm−3 during carbon nanofiber manufacturing operations,18 exceeded 1330 μm2 cm−3 during direct gas-fired heater operation,26 and exceeded 2000 μm2 cm−3 observed during iron foundry pouring.26
Photoelectric response averaged 29 for engine fires overall and 34 for cabin fires overall (arbitrary units). The response provided by the photoelectric aerosol sensor is sensitive to particle adsorbed PAH and would suggest that the overall particle bound PAH concentrations during cabin fires exceeded those from engine fires. However, by normalizing the photoelectric response to the active surface area concentration,27 we obtained ratios of 0.31 for engine fires and 0.11 for cabin fires, suggesting, qualitatively at least, that engine fires were more enriched with particle bound PAH than cabin fires. As the photoelectric response is not calibrated to vehicle combustion aerosol, it is difficult to provide a more quantitative analysis in this instance. The photoelectric response during carbon nanofiber manufacturing operations18 averaged 70, greater than all TWAs obtained during engine or passenger cabin fire suppression (Table 1). However, the maximum photoelectric response of 1400 during the vehicle 2 passenger cabin fire (Table 1) exceeded the maximum of 415 observed during carbon nanofiber manufacturing operations.18
Lönnermark and Blomqvist,4 through similar ELPI measurements to those here, reported a particle size distribution from a vehicle fire with a mode just below 100 nm at maximum number concentration emission. At maximum particle mass emission, the mass size distribution had a sub-micrometer mode, suggesting predominately respirable particles. Our results in Fig. 4 from all six fires show a similar distribution. On a particle number basis, the sub-100 nm or ultrafine particle fraction made up the majority of measured particles throughout all engine compartment and passenger cabin fires, although the fine (100 nm to 1 μm) and coarse fractions (1 μm to 10 μm) also contributed (Fig. 4).
Measured CO concentrations averaged 0.7 ppm and 3.1 ppm from 15 minute TWAs for engine and cabin fires, respectively. Cabin fires were a greater potential source for CO exposure than engine fires with an overall mean of 1.9 ppm obtained for all fires combined. Fent and Evans16 reported a geometric mean of 3.3 ppm CO from the 12 personal breathing zone samples for the fire crews obtained during the same series of vehicle fires, with a maximum transient concentration of 200 ppm. Despite significant differences in the respective sampling approaches, the CO measurements from within the duct and from within the personal breathing zones appear to be similar. However, the maximum transient concentration of CO measured from within the duct (during vehicle 3 cabin fire) was only 62 ppm, below the NIOSH ceiling limit of 200 ppm.28 Exposure to CO reduces systemic transportation and intracellular use of oxygen.29
A majority of on-duty cardiovascular fatalities for firefighters occur during the afternoon or evening hours,30,31 in stark contrast to coronary heart disease deaths in the general population, which tend to peak in the morning hours.32 Kales et al.11 reported a relative risk increase of 60 to 100 times for sudden cardiac fatalities at the fire ground, or shortly following the fire suppression, when compared to non-emergency duties, and the fatality risk increased approximately threefold over physical training alone. Emergency fire response is therefore considered a risk factor for fatality from coronary heart disease.11,33
Short term particle exposures34–36 and potential additive effects with exposure to CO37,38 and short duration, high intensity noise39 may be the environmental factors contributing to adverse cardiovascular events.13 Together with personal risk factors,13 physical exertion40,41 and heat stress,42,43 these combined stressors may be sufficient to trigger adverse cardiac events and warrant further investigation.
In the general population, exposure to ambient fine particles for periods over a few hours to weeks can trigger cardiovascular related mortality and non-fatal events.7 It should be noted that ambient concentrations, even during what are considered high concentration air pollution episodes, are significantly lower than those potentially encountered by firefighters during a fire response. Chronic exposure (e.g. over years) is known to increase the risk for cardiovascular mortality to an even greater extent than exposures over a few days and reduces life expectancy within more highly exposed segments of the population by several months to a few years.7 In addition to cardiovascular-related deaths, chronic exposures to particulate are associated with excess mortality from cancer and respiratory causes.44–46 Furthermore, ultrafine particle deposition within the human respiratory system is increased by almost five fold during exercise.47 On a particle number basis, vehicle fire smoke is substantially ultrafine in nature (Fig. 4 and 5) and physical exertion is an essential component of fire response.48 No short term particle exposure limits are applicable for comparison, but, for example, 15 minute TWAs as we have reported in Table 1 might be useful when examining the relationship with particle exposure and acute adverse cardiac events.
Chemical speciation or compositional analysis of particulate matter contaminants was not conducted during this small study, but specific analytes were monitored in the vapor and gas form.13,14 Previous studies with vehicle fire emissions have reported PAHs, metals, carbonaceous materials, and adsorbed volatiles present in airborne particulate within the smoke from vehicle fires.3,4,25 Some listed contaminants are known or probable carcinogens. Firefighters are at an increased risk of developing certain cancers within the respiratory, digestive, and urinary systems when compared to the general population.8 Combustion aerosols, such as from vehicle fires reported here (Fig. 4 and 5) and elsewhere4 are predominantly respirable in size and are capable of penetrating to the unciliated, gas exchange regions of the lungs, but are deposited throughout the entire respiratory system.49 Particles deposited within the ciliated airways are cleared over time; many of these particles will migrate (be swallowed) into the digestive tract, where constituents may be further metabolized. One study suggests that short-term high intensity particle exposures may be linked to an enhanced cancer risk when compared to lower but longer term particle exposures.50 Thus inhaled particles could conceivably play a role in the increased incidence of certain cancers in firefighters.
Limitations
The flexible metallic duct used in this study enabled direct reading monitoring from a safe distance, but also provided some sampling limitations. Particle losses within the duct were minimized by the high flow rate employed, and consequently, a short contaminant residence time. Nevertheless, for the turbulent flow within the duct (Re ∼60![[thin space (1/6-em)]](https://www.rsc.org/images/entities/char_2009.gif) 000) combined gravitational (∼3%) and inertial deposition losses (∼2%) were estimated at ∼5% for a worst case at 10 μm.51,52 Diffusional particle losses at ∼1.5% were estimated for a worst case at 10 nm.51,52 However, thermophoretic and electrostatic losses were assumed to be negligible with contaminants likely entering the duct at close to ambient temperatures (no significant temperature elevations were observed within the duct) and the duct walls were conductive (aluminum). Therefore, little changes in the overall particle size distributions were anticipated.
000) combined gravitational (∼3%) and inertial deposition losses (∼2%) were estimated at ∼5% for a worst case at 10 μm.51,52 Diffusional particle losses at ∼1.5% were estimated for a worst case at 10 nm.51,52 However, thermophoretic and electrostatic losses were assumed to be negligible with contaminants likely entering the duct at close to ambient temperatures (no significant temperature elevations were observed within the duct) and the duct walls were conductive (aluminum). Therefore, little changes in the overall particle size distributions were anticipated.
Whilst the fire crew mostly suppressed fires from either a southeasterly (engine compartment fires) or southerly (passenger cabin fires) position to the burning vehicle, two of the fire crew (nozzle operator and backup) would temporarily transition to the north side of the vehicle during overhaul for the passenger cabin fires. The duct was not sufficiently long to follow in this scenario, and so the inlet remained close to the southerly side of the vehicle with the additional member of the fire crew. Although held by one of the fire crew, the duct inlet was typically positioned just behind the nozzle operator's shoulder. These positioning limitations would have likely resulted in some degree of particle concentration underestimation, and consequently, concentrations reported here are perhaps conservative estimates of the fire crews' potential for exposure during vehicle fire suppression.
Conclusion
Although relatively short in duration, particle concentrations from vehicle fires may exceed background concentrations by orders of magnitude for a variety of particle metrics (number, respirable mass, active surface area, and photoelectric response). On average, firefighters were potentially exposed to a greater extent during passenger cabin fire suppression than engine compartment fire suppression, by all considered metrics. This finding may be explained by the greater available fuel load and a longer suppression. The wind direction and the relative position of the firefighters to the burning vehicles played a significant role in the firefighters' potential for exposure. Although fire crews generally suppressed vehicle fires from an upwind or smoke clear position, the wind would occasionally veer engulfing fire crews in the smoke plume and resulting in maximum transient exposure events. The findings from this and our earlier article16 suggest that although vehicle fires may be rapidly suppressed, intermittent high intensity exposures to gases, vapors and particles from vehicle fires could potentially contribute to firefighters' risk of certain cancers and adverse cardiovascular events, particularly if SCBA is not worn.Additional studies evaluating firefighter exposures during fire suppression are warranted. Based on the findings from this and from our earlier companion article,16 we recommend that SCBA be worn throughout all phases of the vehicle fire response to reduce the potential of firefighter exposures to hazardous components within fire smoke.
Human subjects
This evaluation was in response to a formal NIOSH health hazard evaluation request and involved standard industrial hygiene approaches. Therefore human subjects/institutional review board approval was not required (42 CFR 85).53Funding
This study was supported by the NIOSH Health Hazard Evaluation Program, Division of Surveillance, Hazard Evaluations and Field Studies and by the Division of Applied Research and Technology.Disclaimers
The findings and conclusions in this report are those of the authors and do not necessarily represent the views of the National Institute for Occupational Safety and Health. Mention of product or company name does not constitute endorsement by the Centers for Disease Control and Prevention.Acknowledgements
The authors are grateful to James Couch, Manny Rodriguez, Chad Dowell and Donald Booher for technical assistance during the study and to Fariba Nourian, Charles Urban and Amanda Harney for photography, videography and health communication assistance. The authors acknowledge Paul Baron (deceased), Thomas Hales and Travis Kubale for numerous insightful discussions and Kevin Dunn, Kenneth Mead and Jerry Kratzer for assistance with the duct and blower system. The authors wish to thank Matthew Dahm, Thomas Hales, John Snawder and the anonymous reviewers at the journal for their technical review and helpful comments with this manuscript. The cooperation and support of the small municipal fire department within SouthWest Ohio, who participated in this evaluation, was greatly appreciated.References
- United States Fire Administration, National Fire Data Center, Fire in the United States 2003–2007, Fifteenth Edition, October 2009, http://www.usfa.fema.gov/downloads/pdf/statistics/fa_325.pdf.
- United States Fire Administration, National Fire Data Center, Highway Vehicle Fires (2008–2010), Topical Fire report Series, vol. 13, issue 11, January 2013, http://www.usfa.fema.gov/downloads/pdf/statistics/v13i11.pdf.
- H. Wichmann, W. Lorenz and M. Bahadir, Release of PCDD/F and PAH during vehicle fires in traffic tunnels, Chemosphere, 1995, 31(2), 2755–2766 CrossRef CAS.
- A. Lönnermark and P. Blomqvist, Emissions from an automobile fire, Chemosphere, 2006, 62, 1043–1056 CrossRef PubMed.
- K. Okamoto, N. Watanebe, Y. Hagimoto, T. Chigira, R. Masano, H. Miura, S. Ochiai, H. Satoh, Y. Tamura, K. Hayano, Y. Maeda and J. Suzuki, Burning behavior of sedan passenger cars, Fire Saf. J., 2009, 44, 301–310 CrossRef PubMed.
- C. S. Baxter, C. S. Ross, T. Fabian, J. L. Borgerson, J. Shawon, P. D. Gandhi, J. M. Dalton and J. E. Lockey, Ultrafine Particle Exposure During Fire Suppression- Is it an Important Contributory Factor for Coronary Heart Disease in Firefighters?, J. Occup. Environ. Med., 2010, 52(8), 791–796 CrossRef PubMed.
- R. D. Brook, et al., Particulate Matter Air Pollution and Cardiovascular Disease, Circulation, 2010, 121, 2331–2378 CrossRef CAS PubMed.
- R. D. Daniels, T. L. Kubale, J. H. Yiin, M. M. Dahm, T. R. Hales, D. Baris, S. H. Zahm, J. J. Beaumont, K. M. Waters and L. E. Pinkerton, Mortality and cancer incidence in a pooled cohort of US firefighters from San Francisco, Chicago and Philadelphia (1950-2009), Occup. Environ. Med., 2014, 71, 388–397 CrossRef PubMed.
- E. Pukkala, J. I. Martinsen, E. Weiderpass, K. Kjaerheim, E. Lynge, L. Tryggvadottir, P. Sparén and P. A. Demers, Cancer incidence among firefighters: 45 years of follow-up in five Nordic countries, Occup. Environ. Med., 2014, 71(6), 398–404 CrossRef PubMed.
- The National Fire Protection Association (NFPA), United States Fire Service Fact Sheet (2014), http://www.nfpa.org/%7E/media/Files/Research/Fact%20sheets/FireServiceFactSheet.pdf.
- S. N. Kales, E. S. Soteriades, C. A. Christophi and D. C. Christiani, Emergency duties and deaths from heart disease among fire fighters in the United States, N. Engl. J. Med., 2007, 356, 1207–1215 CrossRef CAS PubMed.
- M. J. Karter and J. L. Molis, US Firefighter Injuries, 2013, National Fire Protection Association, Quincy MA, 2014 Search PubMed.
- D. L. Smith, D. A. Barr and S. N. Kales, Extreme sacrifice: sudden cardiac death in the US fire service, Extreme Physiology & Medicine, 2013, 2, 6 Search PubMed.
- K. W. Fent, D. E. Evans and J. Couch, Evaluation of Chemical and Particle Exposures during Vehicle Fire Suppression Training, NIOSH HHE Report 2008-0241-3113, 2010 Search PubMed.
- K. W. Fent, J. Eisenberg, D. E. Evans, D. Sammons, S. Robertson, C. Striley, J. Snawder, C. Mueller, V. Kochenderfer, J. Pleil, M. Stiegel and G. Horn, Assessment of Dermal Exposure to Polycyclic Aromatic Hydrocarbons in Firefighters, NIOSH HHE Report 2010-0156-3196, 2013 Search PubMed.
- K. W. Fent and D. E. Evans, Assessing the risk to firefighters from chemical vapors and gases during vehicle fire suppression, J. Environ. Monit., 2011, 13(3), 536–543 RSC.
- A. Peters, H. E. Wichmann, T. Tuch, J. Heinrich and J. Heyder, Respiratory effects are associated with the number of ultrafine particles, Am. J. Respir. Crit. Care Med., 1997, 155(4), 1376–1383 CrossRef CAS PubMed.
- D. E. Evans, B. K. Ku, M. E. Birch and K. H. Dunn, Aerosol Monitoring During Carbon Nanofiber Production: Mobile Direct-Reading Sampling, Ann. Occup. Hyg., 2010, 54(5), 514–531 CrossRef CAS PubMed.
- D. E. Evans, W. A. Heitbrink, T. J. Slavin and T. M. Peters, Ultrafine and respirable particles in an automotive grey iron foundry, Ann. Occup. Hyg., 2008, 52(1), 9–21 CrossRef PubMed.
- The National Fire Protection Association (NFPA), United States Vehicle Fire Trends and Patterns, 2010, http://www.nfpa.org/%7E/media/Files/Research/NFPA%20reports/Vehicles/osvehicle.pdf.
- Code of Federal Regulations (CFR), 29 CFR 190.134. U.S. Government Printing Office, Office of the Federal Register, Washington, DC.
- C. C. Austin, G. Dussault and D. J. Ecobichon, Municipal Firefighter Exposure Groups, Time Spent at Fires and Use of Self-Contained-Breathing-Apparatus, Am. J. Ind. Med., 2001, 40, 683–692 CrossRef CAS PubMed.
- D. M. Bolstad-Johnson, J. L. Burgess, C. D. Crutchfield, S. Storment, R. Gerkin and J. R. Wilson, Characterization of firefighter exposures during fire overhaul, Am. Ind. Hyg. Assoc. J., 2000, 61, 636–641 CAS.
- J. L. Burgess, C. J. Nanson, D. M. Bolstad-Johnson, R. Gerkin, T. A. Hysong, R. C. Lantz, D. L. Sherill, C. D. Crutchfield, S. F. Quan, A. M. Bernard and M. L. Witten, Adverse Respiratory Effects Following Overhaul in Firefighters, J. Occup. Environ. Med., 2001, 43(5), 467–473 CrossRef CAS.
- Underwriters Laboratories, Inc. (UL) (2010): Final Report: Firefighter Exposures to Smoke Particulates, by T. Fabian, J.L. Borgerson, S.I. Kerber, et al. (Report No. 08CA31673), http://site.ul.com/global/documents/offerings/industries/buildingmaterials/fireservice/WEBDOCUMENTS/EMW-2007-FP-02093.pdf.
- W. A. Heitbrink, D. E. Evans, B. K. Ku, A. D. Maynard, T. J. Slavin and T. M. Peters, Relationships among particle number, surface area, and respirable mass concentration in automotive engine manufacturing, J. Occup. Environ. Hyg., 2009, 6(1), 19–32 CrossRef CAS PubMed.
- W. R. Ott and H. C. Siegmann, Using multiple continuous fine particle monitors to characterize tobacco, incense, candle, cooking, wood burning and vehicular sources in indoor, outdoor, and in-transit settings, Atmos. Environ., 2006, 40, 821–843 CrossRef CAS PubMed.
- NIOSH, NIOSH Pocket Guide to Chemical Hazards, ed. M. E. Barsen, U.S. Department of Health and Human Services, Centers for Disease Control and Prevention, National Institute for Occupational Safety and Health (NIOSH), Cincinnati, OH, 2010 Search PubMed.
- A. Ernst and J. D. Zibrak, Carbon monoxide poisoning, N. Engl. J. Med., 1998, 339, 1603–1608 CrossRef CAS PubMed.
- T. Hales, T. Baldwin, K. Sexson and S. Brown, NIOSH Fire Fighter Fatality Investigation and Prevention Program, Paper Presented at the IAFF Redmond Symposium, Honolulu, HI, 1999 Search PubMed.
- S. N. Kales, E. S. Soteriades, S. G. Christoudias and D. C. Christiani, Firefighters and on-duty deaths from coronary heart disease: a case control study, Environ. Health: Global Access Sci. Source, 2003, 2, 14 Search PubMed.
- W. J. Elliott, Cyclic and circadian variations in cardiovascular events, Am. J. Hypertens., 2001, 14, 291S–295S CrossRef.
- NIOSH Alert: Preventing Fire Fighter Fatalities Due to Heart Attacks and Other Sudden Cardiovascular Events [DHHS (NIOSH) Publication No. 2007–133].
- M. Riediker, R. B. Devlin, T. R. Griggs, M. C. Herbst, P. A. Bromberg, R. W. Williams and W. E. Cascio, Cardiovascular effects in patrol officers are associated with fine particulate matter from brake wear and engine emissions, Part. Fibre Toxicol., 2004, 1, 2 CrossRef PubMed.
- A. Peters, S. V. Klot, M. Heier, I. Trentinaglia, A. Hörmann, H. E. Wichmann and H. Löwel, Exposure to Traffic and the Onset of Myocardial Infarction., N. Engl. J. Med., 2004, 351(17), 1721–1730 CrossRef CAS PubMed.
- C. A. Pope III, J. B. Muhlestein, H. T. May, D. G. Renlund, J. L. Anderson and B. D. Horne, Ischemic Heart Disease Events Triggered by Short-Term Exposure to Fine Particulate Air Pollution, Circulation, 2006, 114, 2443–2448 CrossRef PubMed.
- E. N. Allred, E. R. Bleecker, B. R. Chaitman, T. E. Dahms, S. O. Gottlieb, J. D. Hackney, M. Pagano, R. H. Selvester, S. M. Walden and J. Warren, Short-term effects of carbon monoxide exposure on the exercise performance of subjects with coronary artery disease, N. Engl. J. Med., 1989, 321, 1426–1432 CrossRef CAS PubMed.
- E. N. Allred, E. R. Bleecker, B. R. Chaitman, T. E. Dahms, S. O. Gottlieb, J. D. Hackney, M. Pagano, R. H. Selvester, S. M. Walden and J. Warren, Effects of carbon monoxide on myocardial ischemia, Environ. Health Perspect., 1991, 91, 89–132 CrossRef CAS.
- R. L. Tubbs, Noise and hearing loss in firefighting, in Firefighters' Safety and Health, Occupational Medicine State of the Art Reviews, ed. P. Orris, J. Melius and R. M. Duffy, Hanley & Belfus, Inc., Philadelphia, PA, 1995, vol. 10, suppl. 4, pp. 843–856 Search PubMed.
- C. M. Albert, M. A. Mittleman, C. U. Chae, I. M. Lee, C. H. Hennekens and J. E. Manson, Triggering of sudden death from cardiac causes by vigorous exertion, N. Engl. J. Med., 2000, 343, 1355–1361 CrossRef CAS PubMed.
- N. L. Mills, H. Törnqvist, M. C. Gonzalez, E. Vink, S. D. Robinson, S. Söderburg, N. A. Boon, K. Donaldson, T. Sandström, A. Blomberg and D. E. Newby, Ischemic and Thrombotic Effects of Dilute Diesel-Exhaust Inhalation in Men with Coronary Heart Disease, N. Engl. J. Med., 2007, 357(11), 1075–1082 CrossRef CAS PubMed.
- R. Rossi, Firefighting and its influence on the body, Ergonomics, 2003, 46, 1017–1033 CrossRef PubMed.
- D. L. Smith, S. J. Petruzzello and T. S. Manning, The Effect of Strenuous Live-Fire Drills on Cardiovascular and Psychological Responses of Recruit Firefighters, Ergonomics, 2001, 44, 244–254 CrossRef CAS PubMed.
- D. W. Dockery, C. A. Pope, X. Xu, J. D. Spengler, J. H. Ware, M. E. Fay, B. G. Ferris and F. E. Speizer, An Association between Air Pollution and Mortality in Six U.S. Cities, N. Engl. J. Med., 1993, 329(24), 1753–1759 CrossRef CAS PubMed.
- C. Pelucchi, E. Negri, S. Gallus, P. Boffetta, I. Tramacere and C. La Vecchia, Long-term particulate matter exposure and mortality: a review of European epidemiological studies, BMC Public Health, 2009, 9, 453–461 CrossRef PubMed.
- K. A. Miller, D. S. Siscovick, L. Sheppard, K. Shepard, J. H. Sullivan, G. L. Anderson and J. D. Kaufman, Long-Term Exposure to Air Pollution and Incidence of Cardiovascular Events in Women, N. Engl. J. Med., 2007, 365(5), 447–458 CrossRef PubMed.
- C. C. Daigle, D. C. Chalupa, F. R. Gibb, P. E. Morrow, G. Oberdörster, M. J. Utell and M. W. Frampton, Ultrafine particle deposition in humans during rest and exercise, Inhalation Toxicol., 2003, 15, 539–552 CrossRef CAS PubMed.
- D. L. Smith and S. J. Petruzzello, Selected physiological and psychological responses to live-fire drills in different configurations of firefighting gear, Ergonomics, 1998, 41(8), 1141–1154 CrossRef CAS PubMed.
- ICRP, 1994. Human Respiratory Tract Model for Radiological Protection. ICRP Publication 66. Ann. ICRP 24 (1–3).
- N. C. Halmes, S. M. Roberts, J. K. Tolson and C. J. Portier, Reevaluating Cancer Risk estimates for Short-Term Exposure Scenarios, Toxicol. Sci., 2000, 58, 32–42 CrossRef CAS.
- J. E. Brockmann, Sampling and transport of aerosols, in Aerosol Measurement Principles, Techniques, and Applications, ed. P.A. Baron and K. Willeke, Wiley-Interscience, New York, 2001, pp. 143–95 Search PubMed.
- W. C. Hinds, Aerosol Technology: Properties, Behavior and Measurement of Airborne Particles, Wiley-Interscience, New York, 2nd edn, 1999 Search PubMed.
- Code of Federal Regulations (CFR), 42 CFR 85. U.S. Government Printing Office, Office of the Federal Register, Washington, DC.
| This journal is © The Royal Society of Chemistry 2015 |