Triazolo-β-aza-ε-amino acid and its aromatic analogue as novel scaffolds for β-turn peptidomimetics†
Abstract
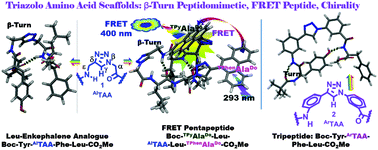
Triazolo-β-aza-ε-amino acid and its aromatic analogue (AlTAA/ArTAA) in the peptide backbone mark a novel class of conformationally constrained molecular scaffolds to induce β-turn conformations. This was demonstrated for AlTAA in a Leu-enkephalin analogue and in a designed pentapeptide wherein the FRET process was established. Restricted rotation induced chirality and turn conformation into the achiral aromatic amino acid scaffold, ArTAA, which in a short tripeptide backbone acted as a β-turn mimic as a β-sheet folding nucleator.
- This article is part of the themed collection: 2015 Emerging Investigators

 Please wait while we load your content...
Please wait while we load your content...