Effects of auxiliary ligands of Pd(ii) dimers on induction of chiral nematic phases: chirality inversion and the photo-responsive structural change†
Abstract
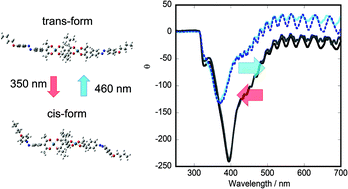
Dinuclear square planar palladium(II) complexes, [{Pd(II)La}2(baet)] (La− = β-diketonato and baet2− = 1,2-diacetyl-1,2-bis(3-methylbutanoyl)ethanato), were prepared and used as chiral dopants to induce chiral nematic phases. The following β-diketones were used as LaH: pentane-2,4-dione (acacH), dibenzoylmethane (dbmH), di-4-nonyloxybenzoylmethane (C9-dbmH) and 3-[4′-(4′′-(octyloxy)phenylazo)phenyl]-2,4-dione (C8-azoacacH). When the enantiomers were doped in a nematic liquid crystal, they induced a chiral nematic phase with a helical twisting power (HTP) of 5–50 μm−1. In particular, the sample doped with [{Pd(II)(C8-azoacac)}2(baet)] exhibited a reversible change of the circular dichroism spectrum under alternate irradiation at 350 nm and 460 nm. It implied that the HTP changed reversibly in response to the cis–trans isomerization of the coordinated C8-azoacac ligand.

 Please wait while we load your content...
Please wait while we load your content...