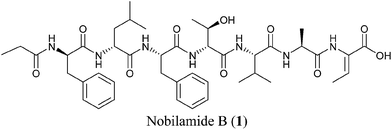
Synthesis and bioactivity of nobilamide B†
M. P. V. Jacintoa,
M. S. Floresb,
Z. Linc,
G. P. Concepcionb,
E. W. Schmidtc,
S. Faulknerd and
A. J. L. Villaraza*a
aInstitute of Chemistry, College of Science, National Science Complex, University of the Philippines, Diliman, Quezon City, 1100, Philippines. E-mail: alvillaraza@up.edu.ph
bMarine Science Institute, College of Science, National Science Complex, University of the Philippines, Diliman, Quezon City, 1100, Philippines
cDepartment of Medicinal Chemistry, University of Utah, Salt Lake City, Utah UT 84112, USA
dDepartment of Chemistry, Chemistry Research Laboratory, University of Oxford, Oxford OX1 3TA, UK
First published on 18th August 2014
Abstract
An alternative and facile solution/solid-phase approach is reported for the total synthesis of neuroactive peptide, nobilamide B. Z-Dhb was formed in solution via EDC/CuCl induced elimination. The solid-phase synthesis employed HBTU/Oxyma Pure™ coupling using Barlos resin. Synthetic nobilamide B was also found to be neuroactive in primary cultures of dorsal root ganglion (DRG) neurons.
Lin et al. reported a family of peptides, coined the nobilamides, which were isolated from Streptomyces sp. cultured from tissues of mollusks, Chicoreus nobilis.1 Nobilamide B (1) exhibited neuroactivity based on its inhibition of the action of capsaicin on TRPV1, implicated in inflammation and sensation of pain.
The synthesis of 1 was first described by Yamashita and co-workers2 using Staudinger ligation to generate the didehydrobutyrine residue in tandem with solid-phase peptide synthesis. In this study, we report an alternative, more flexible and facile synthesis of 1 which can be used for the synthesis of structural analogues.
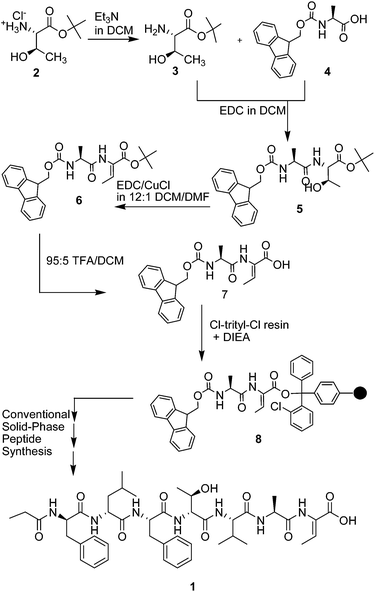
In designing the synthetic strategy to 1, synthesis of the non-proteinogenic amino acid Z-Dhb was first accomplished prior to conventional solid phase synthesis of the complete peptide. Many different approaches to synthesize dehydroamino acids have been reported but elimination is the most described.3 Dehydration of the unprotected activated –OH group is the direct method for the synthesis of dehydroamino acids especially if the β-hydroxy amino acid precursors, such as serine and threonine, are available.4–6 Reports of various reagents used for the activation of the hydroxyl group and elimination with an appropriate base have grown extensively. Jimenez, et al. reported a carbodiimide activation of the hydroxyl group using EDC in the presence of CuCl in both solution and solid-phase methods, where only the Z isomer (the most thermodynamically stable) is formed.5 Almost quantitative conversion is observed using the EDC/CuCl system in the preparation of peptides involving L-Ser, L-Thr (including D-allo-Thr), and L/D-(β-OH)Phe. Didehydroamino acids should not be generated prior to coupling with other amino acids because the carboxylic acid of a didehydroamino acid couples less efficiently than a regular amino acid. Similarly, the presence of the double bond decreases the nucleophilicity of the amine group.5,7
Synthesis of Z-Dhb (Scheme 1) was achieved by the conversion of tert-butyl C-terminal protected threonine, H-Thr-OtBu·HCl (2) to H-Thr-OtBu (3) and coupling with Fmoc-Ala-OH (4) in a carbodiimide based reaction in dichloromethane under basic conditions to produce Fmoc-Ala-Thr-OtBu (5) with 89% yield. Elimination of the –OH of 5 involved a carbodiimide mediated dehydration using EDC in the presence of CuCl via E1 or E1cb mechanism5 similar to the method of preparation of didehydrobutyrine reported for the synthesis of Kahalalide F8 and cyclic lipononadepsipeptide [N-Mst(L-Ser1), D-Ser4, L-Thr6, L-Asp8, L-Thr9]syringotoxin.7
Conversion of 5 to 6 selectively produced the Z-isomer of the alkene with 72% yield. Dehydration reaction can produce either a Z or E isomer or a mixture of both depending on the conditions and reagents. Even though literature sources5 confirm that the reagents used in the dehydration reaction selectively produces the Z-isomer, a 1D NOE experiment (Fig. 1) was performed in order to determine the stereochemical configuration of the double bond. By irradiating the quartet proton signal at 6.73 ppm, which corresponds to Dhb methine proton, the stereochemistry can be confirmed by identifying which of the signals has an increase in intensity.
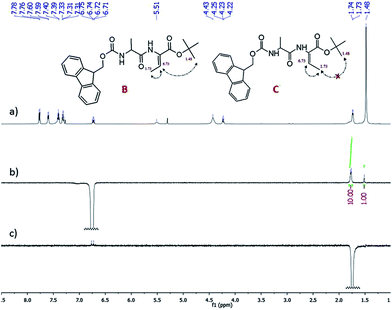 | ||
| Fig. 1 1D NOE Difference spectrum of (6) (a) 1H NMR spectrum, (b) 1D NOE with selective excitation at 6.73 ppm; (c) 1D NOE with selective excitation at 1.73 ppm. | ||
Selective excitation of the proton at 6.73 ppm induces an increase in signal for the proton resonances at 1.73 and 1.48 ppm (Fig. 1b). The increase of the signal in 1.73 ppm corresponds to the interaction of the methine proton with the methyl protons of the Dhb due to their proximity. The increase of the signal at 1.73 ppm is more evident for the methyl protons (ten-fold greater) than the increase in the signal of methyl protons of tert-butyl group at 1.48 ppm because the methyl protons are closer to the methine proton of Dhb relative to the tert-butyl protons. This clearly shows that the methine proton is close to the tert-butyl protons of the C-terminal protecting group due to the increase of signal in 1.48 ppm and verifies that the isomer formed is a Z-isomer.
To further support the claim that the Z-isomer was formed, a 1D NOE spectrum was obtained wherein the selective excitation band chosen was at 1.73 ppm for the tert-butyl protons (Fig. 1c). If the alkene was an E-isomer, irradiating the signal at 1.73 ppm should increase the signal at 1.48 ppm. Clearly, there is no increase in signal observed in the spectrum. Therefore, the stereochemical configuration of the alkene generated is exclusively the Z-isomer.
The formation of Fmoc-Ala-Z-Dhb-OH (7) from 6 involved a simple deprotection of the tert-butyl protecting group of the C-terminal of didehydrobutyrine via AAL1 elimination reaction9 using high concentrations of TFA with 90% yield. The dipeptide 7 successfully conjugated to the Barlos resin using a slight excess of 7 (1.1 equiv.) in the span of 1.5 h. The attachment of the dipeptide was confirmed by the Kaiser test, quantified using Fmoc assay10,11 and calculated to have 1.67 mmol of 7 g−1 of resin (98% loading). After the addition of the dipeptide 7 to the Barlos resin, conventional solid phase peptide synthesis was employed to yield 1 with 92% yield, >40% purity. The peptide 1 was then purified using semi-preparative HPLC before characterization to obtain an overall yield of 25% with >98% purity. The peptide was characterized by 1H NMR (Table 1) and by HRMS with a peak at m/z 858.5299 calculated for the [M + Na]+ (ESI†).
| Residue | Chemical shift, δ ppm (J value, Hz) | ||
|---|---|---|---|
| Isolated | Synthetic | ||
| Z-Dhb | 3 | 6.58 q (7.0) | 6.54 q (7.1) |
| 4 | 1.64 d (7.0) | 1.60 d (7.1) | |
| NH | 9.04 s | 9.00 s | |
| L-Ala | 2 | 4.43 m | 4.38 m |
| 3 | 1.31 d (7.0) | 1.27 d (7.1) | |
| NH | 8.19 d (7.7) | 8.15 d (7.1) | |
| L-Val | 2 | 4.30 dd (6.5, 8.5) | 4.25 m |
| 3 | 2.03 m | 1.99 m | |
| 4/5 | 0.86 d (6.0)/0.88 d (6.0) | 0.82 d (6.0)/0.84 d (6.0) | |
| NH | 7.75 d | 7.70 d | |
| D-a-Thr | 2 | 4.36 dd (7.0, 7.2) | 4.30 m |
| 3 | 3.83 m | 3.79 m | |
| 4 | 1.07 d (5.8) | 1.02 d (6.3) | |
| NH | 8.14 d (8.6) | 8.08 d (8.5) | |
| L-Phe | 2 | 4.59 m | 4.58 m |
| 3 | 3.06 dd/2.71 m | 3.08 dd/2.70 m | |
| Ph | 7.02–7.30 m | 7.14–7.27 m | |
| NH | 8.28 d (8.4) | 8.29 d (8.5) | |
| D-Leu | 2 | 4.20 m | 4.19 m |
| 3 | 1.15 m | 1.15 m | |
| 4 | 1.16 m | 1.16 m | |
| 5/6 | 0.73 d (5.6)/0.70 d (5.6) | 0.73 d (5.9)/0.70 d (6.0) | |
| NH | 7.92 d (8.0) | 7.94 d (8.0) | |
| D-Phe | 2 | 4.48 m | 4.48 m |
| 3 | 2.93 dd/2.69 m | 2.94 dd/2.70 m | |
| Ph | 7.0–7.3 m | 7.14–7.27 m | |
| NH | 7.91 d (8.0) | 7.93 d (8.3) | |
| Prop. | 2 | 1.98 m | 1.99 m |
| 3 | 0.82 m | 0.82 m | |
The isolated and synthetic 1 were co-eluted and the resulting chromatogram showed only a single peak (ESI†). Hence, the solution behaviour of the synthetic peptide is identical with that of the isolated.
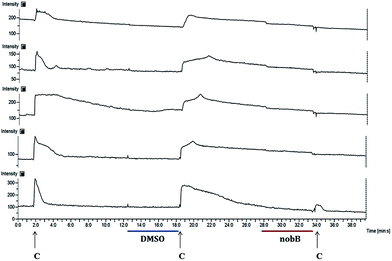
The bioactivity of the synthetic compound was evaluated in the DRG assay. The primary cultures of DRG contain at least 12 definable cell types that express distinct receptor and channel populations, including a substantial fraction of nociceptors (∼30–50%).12–15 Using calcium imaging, we routinely utilize this broad scope assay to screen for neuroactive compounds.
The assay result showed that synthetic nobilamide B inhibited the response of DRG neurons to capsaicin (Fig. 2). This is similar to the result obtained with the isolated natural product, which induced an inhibitory effect to TRPV1.
Conclusions
Nobilamide B (1) was successfully synthesized and found to be structurally identical to the isolated natural product. 1H- and 13C-NMR experiments validate the stereochemical assignment (absolute configuration) of the natural product. HPLC col-elution demonstrates that the synthetic and isolated peptides have the same solution behaviour. Furthermore, the DRG assay demonstrates that the synthetic compound is neuroactive with inhibitory effect on capsaicin. At present, we are employing the synthetic route described above for the production of analogues in order to conduct a comprehensive structure–activity relationship study.Acknowledgements
This research is supported by Philippine Mollusk Symbiont International Cooperative Biodiversity Group (PMS-ICBG, NIH 1U01TW008163-01 “Diverse drug lead compounds from bacterial symbionts in tropical marine mollusks”), International Foundation for Science (IFS Grant no. F-5189), and Office of Vice Chancellor for Research and Development (OVCRD Project no. 121207 TNSE). Special thanks to Louie Carpio and Joshua Torres for generating the ESI-MS data.Notes and references
- Z. Lin, C. A. Reilly, R. Antemano, R. W. Hughen, L. Marett, G. P. Concepcion, M. G. Haygood, B. M. Olivera, A. Light and E. W. Schmidt, J. Med. Chem., 2011, 54, 3746–3755 CrossRef CAS PubMed.
- T. Yamashita, H. Matoba, T. Kuranaga and M. Inoue, Tetrahedron, 2014, 1–7 Search PubMed , in press.
- P. M. T. Ferreira, H. L. S. Maia and L. S. Monteiro, Tetrahedron Lett., 1998, 39, 9575–9578 CrossRef CAS.
- U. Kazmaier, Synthesis and Chemistry of Modified Amino Acids, WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim, 2009, vol. 2 Search PubMed.
- J. C. Jimenez, N. Bayo, B. Chavarria, A. Lopez-Macia, M. Royo, E. Nicolas, E. Giralt and F. Albericio, Lett. Pept. Sci., 2003, 9, 135–141 Search PubMed.
- R. Ramesh, K. De and S. Chandrasekaran, Tetrahedron, 2007, 63, 10534–10542 CrossRef CAS PubMed.
- N. Bayo, J. C. Jimenez, L. Rivas, E. Nicolas and F. Albericio, Chem.–Eur. J., 2003, 9, 1096–1103 CrossRef CAS PubMed.
- A. López-Macià, J. Jiménez, M. Royo, E. Giralt and F. Albericio, J. Am. Chem. Soc., 2001, 123, 11398–11401 CrossRef PubMed.
- R. Bruckner, Organic Mechanisms, Springer-Verlag, Berlin Heidelberg, 2010, 3rd English Search PubMed.
- M. Amblard, J. Fehrentz, J. Martinez and G. Subra, Mol. Biotechnol., 2006, 33, 239–254 CrossRef CAS PubMed.
- M. Gude, J. Ryf and P. D. White, Lett. Pept. Sci., 2003, 9, 203–206 CrossRef PubMed.
- O. Peraud, J. Biggs, R. Hughen, A. Light, G. Concepcion, B. Olivera and E. Schmidt, Appl. Environ. Microbiol., 2009, 75, 6820 CrossRef CAS PubMed.
- A. Light, R. Hughen, J. Zhang, J. Rainier, Z. Liu and J. Lee, J. Neurophysiol., 2008, 100, 1184 CrossRef CAS PubMed.
- R. Teichert, S. Raghuraman, T. Memon, J. Cox, T. Foulkes, J. Rivier and B. Olivera, Proc. Natl. Acad. Sci. U. S. A., 2012, 109, 12758 CrossRef CAS PubMed.
- R. Teichert, N. Smith, S. Raghuraman, D. Yoshikami, A. Light and B. Olivera, Proc. Natl. Acad. Sci. U. S. A., 2012, 109, 1388 CrossRef CAS PubMed.
Footnote |
| † Electronic supplementary information (ESI) available: Analytical HPLC profile, NMR and ESI-mass spectra are included in the supplementary information. See DOI: 10.1039/c4ra06873d |
| This journal is © The Royal Society of Chemistry 2014 |