Bioimaging based on fluorescent carbon dots
Abstract
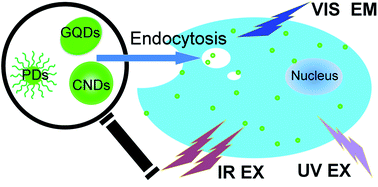
Nanosized fluorescent carbon particles, namely, carbon dots (CDs), are a kind of fluorescent material that has drawn increasing attention in recent years. CDs have size-, surface chemistry-, and wavelength-dependent luminescence emission, which is different from traditional semiconductor-based quantum dots. Moreover, with excellent chemical stability, good biocompatibility, low toxicity, up-conversion emission, resistance to photo bleaching, as well as easy chemical modifications, CDs are promising for substantial applications in numerous areas: bioimaging, sensors, and energy-related devices. Herein, three kinds of fluorescent dots are reviewed: graphene quantum dots (GQDs), carbon nanodots (CNDs) and polymer dots (PDs). After the first reported CDs prepared from electrophoretic analysis and purification of fluorescent carbon nanotube fragments, there were hundreds of publications focusing on fluorescent CDs. Bioimaging was one of the most common applications of the CDs; therefore, in this review, most of the chosen reference papers were related to bioimaging based on CDs.
- This article is part of the themed collections: Chemistry for Medicine: Special Collection for RSC Advances and Cellular and Tissue Imaging – Luminescent Tags and Probes

 Please wait while we load your content...
Please wait while we load your content...