Enantioselective imine reduction catalyzed by imine reductases and artificial metalloenzymes
Abstract
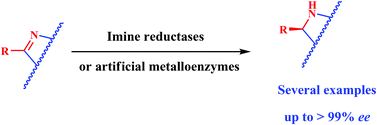
Adding value to organic synthesis. Novel imine reductases enable the enantioselective reduction of imines to afford optically active amines. Likewise, novel bioinspired artificial metalloenzymes can perform the same reaction as well. Emerging proof-of-concepts are herein discussed.

 Please wait while we load your content...
Please wait while we load your content...