Abstract
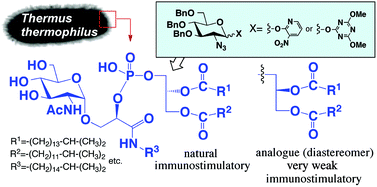
An extreme thermophile, Thermus thermophilus, has very unique glycolipids on the cell surface. The acidic immunostimulatory phosphoglycolipid of T. thermophilus was synthesized for the first time, with newly developed glycosylation methods using 3-nitropyridyl (3NPy) and 4,6-dimethoxy-1,3,5-triazin-2-yl (DMT) glycosides as glycosyl donors. The analogues of the phosphoglycolipid, which include a diastereomer possessing the opposite configuration at the diacyl glycerol moiety, were also synthesized. The biological activities of the synthesized compounds were elucidated with cytokine inductions (IL-6 and TNF-α). A synthetic phosphoglycolipid with a natural-type diacyl glycerol configuration showed apparent immunostimulatory activity, whereas its diastereomer did not. The present study revealed that the configuration at the diacyl glycerol moiety of the phosphoglycolipids is important for immunostimulation, suggesting the existence of the particular receptor/recognizing protein that can recognize the stereochemistry of the glycerol part.
- This article is part of the themed collection: In Celebration of Andrew D. Hamilton’s Career in Chemistry

 Please wait while we load your content...
Please wait while we load your content...