Mesoporous α-Fe2O3 thin films synthesized via the sol–gel process for light-driven water oxidation†
Abstract
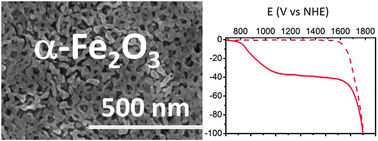
This work reports a facile and cost-effective method for synthesizing photoactive α-Fe2O3 films as well as their performances when used as photoanodes for water oxidation. Transparent α-Fe2O3 mesoporous films were fabricated by template-directed sol–gel chemistry coupled with the dip-coating approach, followed by annealing at various temperatures from 350 °C to 750 °C in air. α-Fe2O3 films were characterized by X-ray diffraction, XPS, FE-SEM and electrochemical measurements. The photoelectrochemical performance of α-Fe2O3 photoanodes was characterized and optimized through the deposition of Co-based co-catalysts via different methods (impregnation, electro-deposition and photo-electro-deposition). Interestingly, the resulting hematite films heat-treated at relatively low temperature (500 °C), and therefore devoid of any extrinsic dopant, achieve light-driven water oxidation under near-to-neutral (pH = 8) aqueous conditions after decoration with a Co catalyst. The onset potential is 0.75 V vs. the reversible hydrogen electrode (RHE), thus corresponding to 450 mV light-induced underpotential, although modest photocurrent density values (40 μA cm−2) are obtained below 1.23 V vs. RHE. These new materials with a very large interfacial area in contact with the electrolyte and allowing for a high loading of water oxidation catalysts open new avenues for the optimization of photo-electrochemical water splitting.

 Please wait while we load your content...
Please wait while we load your content...