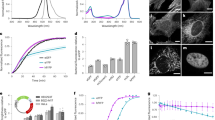
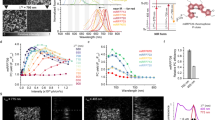
Abstract
Infrared fluorescent proteins (IFPs) provide an additional color to GFP and its homologs in protein labeling. Drawing on structural analysis of the dimer interface, we identified a bacteriophytochrome in the sequence database that is monomeric in truncated form and engineered it into a naturally monomeric IFP (mIFP). We demonstrate that mIFP correctly labels proteins in live cells, Drosophila and zebrafish. It should be useful in molecular, cell and developmental biology.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
Accession codes
Primary accessions
NCBI Reference Sequence
Referenced accessions
NCBI Reference Sequence
Protein Data Bank
References
Day, R.N. & Davidson, M.W. Chem. Soc. Rev. 38, 2887–2921 (2009).
Tsien, R.Y. Angew. Chem. Int. Ed. Engl. 48, 5612–5626 (2009).
Shu, X. et al. Science 324, 804–807 (2009).
Filonov, G.S. et al. Nat. Biotechnol. 29, 757–761 (2011).
Auldridge, M.E., Satyshur, K.A., Anstrom, D.M. & Forest, K.T. J. Biol. Chem. 287, 7000–7009 (2012).
Shcherbakova, D.M. & Verkhusha, V.V. Nat. Methods 10, 751–754 (2013).
Piatkevich, K.D., Subach, F.V. & Verkhusha, V.V. Nat. Commun. 4, 2153–2162 (2013).
Giraud, E. & Verméglio, A. Photosynth. Res. 97, 141–153 (2008).
Rockwell, N.C., Su, Y.S. & Lagarias, J.C. Annu. Rev. Plant Biol. 57, 837–858 (2006).
Karniol, B., Wagner, J.R., Walker, J.M. & Vierstra, R.D. Biochem. J. 392, 103–116 (2005).
Yu, D. et al. Nat. Commun. 5, 3626–3632 (2014).
Geiler-Samerotte, K.A. et al. Proc. Natl. Acad. Sci. USA 108, 680–685 (2011).
Wagner, J.R., Brunzelle, J.S., Forest, K.T. & Vierstra, R.D. Nature 438, 325–331 (2005).
Stemmer, W.P. Nature 370, 389–391 (1994).
Szymczak, A.L. et al. Nat. Biotechnol. 22, 589–594 (2004).
Cui, L. et al. Biochem. Biophys. Res. Commun. 377, 1156–1161 (2008).
Han, C., Jan, L.Y. & Jan, Y.-N. Proc. Natl. Acad. Sci. USA 108, 9673–9678 (2011).
Shaner, N.C. et al. Nat. Biotechnol. 22, 1567–1572 (2004).
Shemiakina, I.I. et al. Nat. Commun. 3, 1204–1207 (2012).
To, T.-L. et al. Proc. Natl. Acad. Sci. USA 112, 3338–3343 (2015).
Acknowledgements
This work was supported by the Program for Breakthrough Biomedical Research (to X.S.); US National Institute of Health (NIH) GM030637 (to T.B.K.), GM084040 and GM096164 (to O.D.W.); and the Howard Hughes Medical Institute (to Y.-N.J.). S.-Q.Z. was supported by NIH GM054616 (to W.F. DeGrado) and US National Science Foundation DMR-1120901. We thank N. Joh for assistance in gel filtration chromatography; A. Royant for size-exclusion chromatography; W.F. DeGrado for providing access to the analytical ultracentrifuge; L.D. Wilsbacher, A. Schepis and S.R. Coughlin for initial testing of mIFP in zebrafish; C.S. Craik for constructive comments; S. Woo at University of California, San Francisco (UCSF) for providing the pCS2+ vector; and S. Roy (UCSF) for providing the UAS-mCherry Drosophila line.
Author information
Authors and Affiliations
Contributions
X.S. conceived the project. D.Y. and X.S. designed mIFP and the H2B fusion. K.M. and X.S. planned the Drosophila embryo imaging. M.W.D. planned the fusion constructs. T.B.K. planned the histone H3.3 fusion construct and the transgenic Drosophila. A.R. and O.D.W. planned the imaging of zebrafish. M.P.K., Y.S. and Y.-N.J. planned the imaging of epithelia, muscle and neurons in Drosophila larvae and adults. D.Y., M.A.B., J.R.A., E.S.H., M.P.K., A.R., K.M., Y.S., S.L., Z.M. and S.-Q.Z. performed the experiments. D.Y. and X.S. wrote the manuscript. All the authors contributed to the final draft.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–21 and Supplementary Tables 1 and 2 (PDF 29544 kb)
Z-section confocal imaging of mIFP-α-tubulin in live cells.
HEK293 cells transiently transfected with the fusion construct were imaged with z-section (step size 1 μm). The entire field of view is shown. (AVI 1664 kb)
Time lapse imaging of mIFP-α-tubulin in live cells.
HEK293 cells transiently transfected with the fusion construct were imaged every 15 min. The entire field of view is shown. (AVI 2039 kb)
Time lapse imaging of mIFP-β-actin in live cells.
Ptk2 cells transiently transfected with the fusion construct were imaged every 3 min. (AVI 2222 kb)
Time lapse imaging of mIFP-EB3 in live cells.
HeLa cells transiently transfected with the fusion construct were imaged every 2 seconds. (AVI 438 kb)
Time lapse imaging of mIFP-H1 in live cells.
HeLa cells transiently transfected with the fusion construct were imaged every 2 min. (AVI 3536 kb)
Z-section two-color confocal imaging.
HEK293 cells transiently transfected with the mRuby-H2B (in yellow) and mIFP-α-tubulin (in red) were imaged with z-section (step size 1 μm). (AVI 125 kb)
Time lapse imaging of CD8-GFP in Drosophila.
Entire Drosophila embryo expressing UAS-CD8-GFP driven by elav-GAL4 was imaged every 10 min, which revealed the ventral nerve cord condensation. (AVI 502 kb)
Time lapse imaging of mIFP-H3.3 T2A HO1 and CD8-GFP in Drosophila.
Entire Drosophila embryo expressing UAS-mIFP-H3.3 T2A HO1 (in red) and UAS-CD8-GFP (in green) driven by elav-GAL4 was imaged every 10 min, which revealed the ventral nerve cord condensation. (AVI 531 kb)
Rights and permissions
About this article
Cite this article
Yu, D., Baird, M., Allen, J. et al. A naturally monomeric infrared fluorescent protein for protein labeling in vivo. Nat Methods 12, 763–765 (2015). https://doi.org/10.1038/nmeth.3447
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.3447
This article is cited by
-
Quantitative assessment of near-infrared fluorescent proteins
Nature Methods (2023)
-
Deep-tissue SWIR imaging using rationally designed small red-shifted near-infrared fluorescent protein
Nature Methods (2023)
-
Polarized focal adhesion kinase activity within a focal adhesion during cell migration
Nature Chemical Biology (2023)
-
Optogenetic manipulation and photoacoustic imaging using a near-infrared transgenic mouse model
Nature Communications (2022)
-
Long-term monitoring of intravital biological processes using fluorescent protein-assisted NIR-II imaging
Nature Communications (2022)