Vacuum-evaporable spin-crossover complexes: physicochemical properties in the crystalline bulk and in thin films deposited from the gas phase†‡
Abstract
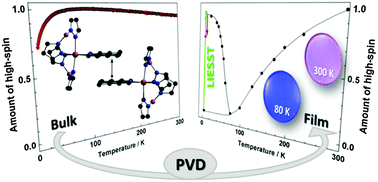
Four analogues of the spin-crossover complex [Fe(H2Bpz2)2(phen)] (H2Bpz2 = dihydrobis(pyrazolyl)borate; 2) containing functionalized 1,10-phenanthroline (phen) ligands have been prepared; i.e., [Fe(H2Bpz2)2(L)], L = 4-methyl-1,10-phenanthroline (3), 5-chloro-1,10-phenanthroline (4), 4,7-dichloro-1,10-phenanthroline (5), and 4,7-dimethyl-1,10-phenanthroline (6). The systems are investigated by magnetic susceptibility measurements and a range of spectroscopies in the solid state and in thin films obtained by physical vapour deposition (PVD). Thermal as well as light-induced SCO behaviour is observed for 3–6 in the films. By contrast, thermal SCO in the solid state occurs only for 3 and 4 but is absent for 5 and 6. These findings are discussed in the light of cooperative and intermolecular interactions.
- This article is part of the themed collection: Spin State Switches in Molecular Materials Chemistry

 Please wait while we load your content...
Please wait while we load your content...