Redox-controlled release of molecular payloads from multilayered organometallic polyelectrolyte films†
Abstract
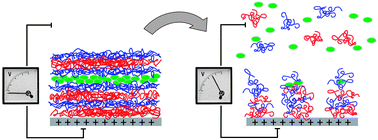
Organometallic poly(ferrocenylsilane)s (PFSs) featuring redox responsive ferrocene units in their main chain are prepared and characterized for redox triggered film disassembly and release of molecular payloads. Positively or negatively charged side groups render PFS

 Please wait while we load your content...
Please wait while we load your content...