A precious metal-free solar water splitting cell with a bifunctional cobalt phosphide electrocatalyst and doubly promoted bismuth vanadate photoanode†
Abstract
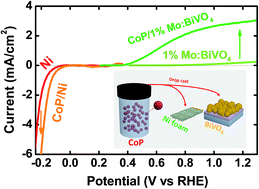
A bifunctional cobalt phosphide (CoP) electrocatalyst is applied to a doubly promoted BiVO4 photoanode as an oxygen evolution as well as to a cathode as a hydrogen evolution reaction (HER) catalyst to establish a photoelectrochemical (PEC) water splitting cell made of only earth abundant elements without any precious metals. Although the intrinsic HER activity of CoP is lower than Pt/C, CoP can replace Pt as the cathode of PEC water splitting cells without any significant loss in performance. During the water splitting reaction, the cathode remains as CoP, but CoP loaded on the BiVO4 photoanode turns into amorphous CoOx–HPOy with its chemical state and performance very similar to those of well-known cobalt phosphate electrocatalysts. A tandem cell composed of CoP/hydrogen-treated, 1% Mo-doped BiVO4 as the photoanode, a CoP/Ni foam as the cathode and a 2-piece, series connected crystalline Si solar cell as the bias power generator demonstrates stable unassisted water splitting with a solar-to-hydrogen conversion efficiency of 5.3% under simulated solar light, which represents the highest among reported precious metal-free unassisted solar water splitting devices.


 Please wait while we load your content...
Please wait while we load your content...