Structure–property insights into nanostructured electrodes for Li-ion batteries from local structural and diffusional probes†
Abstract
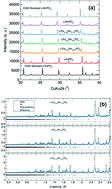
Microwave heating presents a faster, lower energy synthetic methodology for the realization of functional materials. Here, we demonstrate for the first time that employing this method also leads to a decrease in the occurrence of defects in olivine structured LiFe1−xMnxPO4. For example, the presence of antisite defects in this structure precludes Li+ diffusion along the b-axis leading to a significant decrease in reversible capacities. Total scattering measurements, in combination with Li+ diffusion studies using muon spin relaxation (μ+SR) spectroscopy, reveal that this synthetic method generates fewer defects in the nanostructures compared to traditional solvothermal routes. Our interest in developing these routes to mixed-metal phosphate LiFe1−xMnxPO4 olivines is due to the higher Mn2+/3+ redox potential in comparison to the Fe2+/3+ pair. Here, single-phase LiFe1−xMnxPO4 (x = 0, 0.25, 0.5, 0.75 and 1) olivines have been prepared following a microwave-assisted approach which allows for up to 4 times faster reaction times compared to traditional solvothermal methods. Interestingly, the resulting particle morphology is dependent on the Mn content. We also examine their electrochemical performance as active electrodes in Li-ion batteries. These results present microwave routes as highly attractive for reproducible, gram-scale syntheses of high quality nanostructured electrodes which display close to theoretical capacity for the full iron phase.


 Please wait while we load your content...
Please wait while we load your content...