Platinum nanoparticles partially-embedded into carbon sphere surfaces: a low metal-loading anode catalyst with superior performance for direct methanol fuel cells†
Abstract
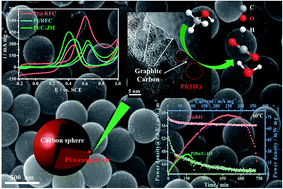
Pt-based catalysts are considered as the most efficient and indispensable catalysts for methanol electro-oxidation reactions (MORs) in acidic media; however, issues linked to cost and stability impede their large-scale application. Here, we present a novel structured catalyst with Pt nanoparticles partially embedded in resorcinol-formaldehyde carbon spheres (Pt@RFC) towards MORs. Pt@RFC exhibits excellent CO-tolerance and MOR activity, and specifically, the CO electro-oxidation peak-potential is negatively shifted by ∼150 mV and the electrocatalytic activity is 2 times that of commercial Pt/C. These enhancements are due to the endowed high Pt utilization (decreased particle size) from strong metal-support interaction and the decorated electronic properties. Moreover, the firmly anchored Pt nanoparticles are prevented from possible dissolution, agglomeration and detachment during long-term use. Remarkably, after an accelerated degradation test through a 3000 cycle cyclic voltammetry test, the mass activity for Pt@RFC is well maintained and 5.8 times that of the commercial Pt/C. Upon integration into a DMFC, Pt@RFC (58.5 mW cm−2) exhibits a competitive power density at 60 °C compared to a commercial PtRu/C catalyst (52.3 mW cm−2) with only 1/3 of the noble metal loading, as well as a slower degradation rate during discharge testing. The present findings indicate that Pt@RFC might be a viable alternative as a commercial catalyst applied in DMFCs.


 Please wait while we load your content...
Please wait while we load your content...