Applications and limitations of two step metal oxide thermochemical redox cycles; a review
Abstract
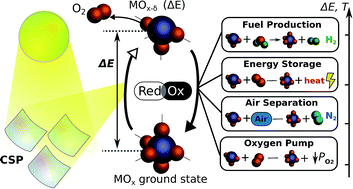
Two step metal oxide thermochemical redox cycles have seen growing interest in the research community over the last two decades. In particular, they have often been studied as a means of converting heat from concentrated solar power to chemical energy, which can subsequently be used for a number of applications. In this work, we offer critical perspective and valuable insight on these research fields, from authors with a combined experience of 50+ years in metal oxide redox cycles. The current fundamental understanding of thermochemical redox materials, and the implications and limitations this has on the redox thermodynamics are discussed. The underlying fundamentals of the redox materials are then used to give insight into the theoretical limitations imposed on a number of applications including; solar thermochemical fuel production, solar energy storage for off sun power generation, thermochemical air separation, oxygen pumping, and thermochemical heat pumping. A number of recent novel process developments are also presented, which offer valuable motivation and direction for the respective fields. The analysis shows that oxides which undergo a stoichiometric phase change during reduction, such as ZnO or Co3O4, have much larger specific energy storage than materials undergoing partial reduction such as ceria and perovskites. The partial reduction materials generally have faster kinetics and better activity at low temperature, and the selection of materials for the various applications is often a compromise between the importance of high specific energy storage vs. fast kinetics and low temperature operation.
- This article is part of the themed collection: Recent Review Articles


 Please wait while we load your content...
Please wait while we load your content...