3D hierarchical CuO mesocrystals from ionic liquid precursors: towards better electrochemical performance for Li-ion batteries†
Abstract
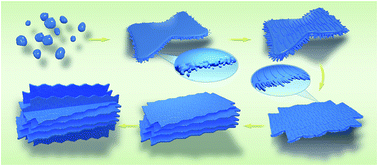
CuO mesocrystals have been synthesized by nonclassical crystallization in the presence of an ionic liquid and n-butylamine under hydrothermal conditions. The resultant mesocrystals are composed of anisotropic nanosheets and nanorods as building blocks and possess distinct 3D hierarchical superstructure exposed {001} crystal planes. The mechanisms underlying the sequential formation of the mesocrystals are as follows: amorphous particles first appeared under a high degree of supersaturation according to the Ostwald rule of stages; then, nanosheet subunits were favored to form due to the protection of the {001} planes by n-butylamine molecules and etching of the {010} planes by a hydrolysis reaction, leading to the formation of a large number of dangling bonds in the {010} planes. Therefore, the ionic liquid can interact with these primary particles to facilitate a self-assembled superstructure by π–π interactions along the [010] direction due to the highly ionic nature, resulting in a 3D framework structure of primary particles composed of nanosheet and nanorod subunits by oriented attachment. Moreover, owing to the inherent porosity associated with well-defined nanoparticle orientation, the 3D hierarchical CuO mesocrystals achieved a higher electrochemical property as anodes for Li-ion batteries, surpassing the performance of CuO nanosheets and CuO nanorods. The unique characteristics of the hierarchical mesostalline electrodes show an ideal geometry to form a stable SEI film, which offers a facile route for designing high-performance electrodes for long-life Li-ion batteries.

 Please wait while we load your content...
Please wait while we load your content...