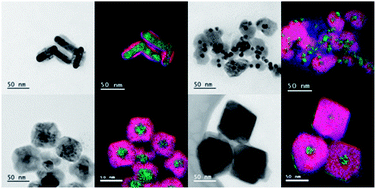
We studied the chemical, optical and catalytic properties of metal (Pt, Pd, Ag, Au)–Cu2O core–shell nanoparticles grown via a simple and reproducible approach which utilizes aqueous-phase reactions at room temperature. We were able to control the thickness of the Cu2O shell and examine the effect of the core's shape and size on the structure and properties of the hybrid nanocrystals. We also studied the optical properties of the hybrid nanocrystals, in particular the effect of the Cu2O shell thickness on the frequency of the plasmon of gold nanorods. In addition, the catalytic activity of the hybrid nanostructures was examined by testing the reduction reaction of 4-nitrophenol with NaBH4. Finally, the hybrid metal–Cu2O nanostructures were used as templates to form the yolk–shell of metal–Cu2S materials. The interface and the crystalline structures of the four hybrid nanostructures were extensively characterized by high resolution transmission electron microscopy (HRTEM), energy-filtered TEM (EFTRM) and X-ray diffraction (XRD).
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...