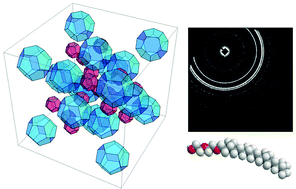
The phase behaviour (both neat and lyotropic) and toxicity of eight new ethylene oxide amphiphiles (EO = 1 to 8) with a single phytanyl chain (3,7,11,15 tetramethylhexadecyl) is reported. There is a discontinuity at EO > 4 where the neat and lyotropic behaviour exhibit a tipping point which is qualitatively rationalised in terms of the molecular geometry of the surfactant. Below four EO units the behaviour of the neat surfactants show only a glass transition, Tg ∼ −90 °C. Above four EO units crystallisation (Tcrys) and crystal-isotropic liquid (Tm) transitions are also observed. These increase monotonically with the hydrophilicity of the surfactant; consistent with the greater cohesiveness of the molecules due to van der Waals interactions. The increase in hydrophilicity corresponds to a decrease in curvature of the surfactant layer towards water. However, the exaggerated splay of the phytanyl chain is effective in promoting various self-assembled structures with inverse cubic and hexagonal phases preferred below ambient temperatures for EO < 4, and these are stable to dilution. Variation of the EO head group length promotes an interesting diversity of cubic phases, with an inverse micellar cubic phase (Fd3m) present for EO = 2 and the bicontinuous gyroid cubic (Ia3d) and double diamond cubic (Pn3m) phases present at higher ethoxylation. DIT-NIR microspectroscopy provided a high throughput, low volume, fast equilibrating method for obtaining the approximate partial temperature-composition phase diagrams of the binary systems with water. The toxicity of colloidal dispersions of these amphiphiles was assayed against normal breast epithelial (HMEpiC) and breast cancer (MCF7) cell lines. The IC50 of the EO amphiphiles was similar in both cell lines with moderate toxicity ranging from ∼80–110 μM in an in vitro cell viability assay.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...