Ion association in low-polarity solvents: comparisons between theory, simulation, and experiment
Abstract
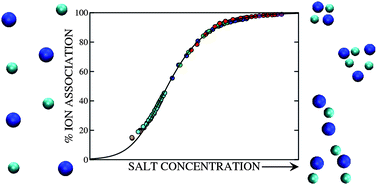
The association of ions in electrolyte solutions at very low concentration and low temperature is studied using computer simulations and quasi-chemical ion-pairing theory. The specific case of the restricted primitive model (charged hard spheres) is considered. Specialised simulation techniques are employed that lead to efficient sampling of the arrangements and distributions of

 Please wait while we load your content...
Please wait while we load your content...