Electrochemical probing of carbon quantum dots: not suitable for a single electrode material
Abstract
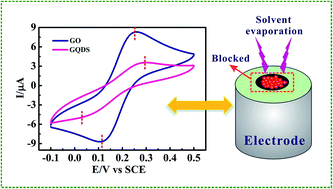
In this article graphene quantum dots (GQDs) were prepared from activated graphene oxide (AGO) for the first time utilizing a reflux route. We focus on the relationship between surface structure and the electrochemical activity of graphene oxide (GO), AGO, GQDs and reduced graphene quantum dots (RGQDs). Surprisingly it was found that GQDs exhibited a much slower electron transfer rate of 1.105 × 10−4 cm s−1 compared with GO of 1.918 × 10−3 cm s−1 and AGO of 6.316 × 10−3 cm s−1. Obviously, a GQDs modified electrode surface can be completely blocked which might result from its low edge-to-basal plane rate though easy aggregation, rapid stacking and high oxygen-functional groups. Contrary to a general view that size-reducing is favorable to electrochemical kinetics, such findings, never reported before, imply that GQDs may not be suitable for applying to a beneficial single electrode material. GQDs, a new type of advanced materials of nanoscale, must be applied in a proper way in order to exert potential superiority to an utmost extent.

 Please wait while we load your content...
Please wait while we load your content...