First-principles investigation on the structural, electronic properties and diffusion barriers of Mg/Al doped NaCoO2 as the cathode material of rechargeable sodium batteries
Abstract
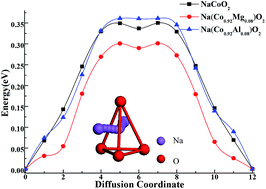
Mg/Al doped NaCoO2 layered transition-metal oxides as potential cathode materials for sodium ion batteries have been investigated by first-principle calculations. The effects of divalent Mg ion or trivalent Al ion doping on the crystal structure, electron transfer, the changes of valence, average intercalation voltage and diffusion barriers of NaCoO2 are studied. The DFT calculations indicate NaCoO2 with Mg or Al ions doping will lead to a higher average intercalation voltage, which is beneficial for obtaining high energy density. Charge disproportionation induced by divalent Mg ion doping results in the appearance of electronic holes in Na(Co0.92Mg0.08)O2, which may enhance its conductivity significantly. The nudged elastic band calculation results indicate that trivalent Al ion doping has a slight effect on the diffusion barriers of NaCoO2, but divalent Mg ion doping can significantly decrease the diffusion barriers and enhance the Na ion diffusion rate, which is beneficial to the improvement of the rate capability.

 Please wait while we load your content...
Please wait while we load your content...