Accurate measurement of stiffness of leukemia cells and leukocytes using an optical trap by a rate-jump method†
Abstract
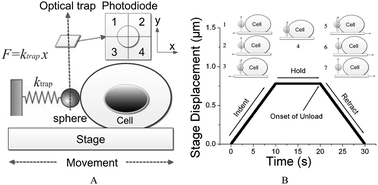
Accurate measurement of the elastic modulus of soft biological cells in the micro/nano scale range is still a challenging task. Tests involving constant-rate loading often yield results that are rate dependent, due to the viscous component of the deformation. In this work, a rate-jump indentation method was employed in an optical tweezers system to measure the stiffness of non-adherent blood cells, which are the softest types of cells. Compared to the traditional Hertzian method of indentation, the rate-jump method is found to be able to yield invariant elastic modulus from K562 myelogenous leukemia cells. The optical tweezers indentation method proposed can therefore serve as a standard protocol for obtaining the intrinsic elastic modulus of extremely soft cells, with applied forces in the pico-newton range. This method is also found to be effective in grading the stiffness values of myelogenous leukemia cell lines (K562 and HL60) and normal leukocytes, indicating that it can be used to identify normal cells from diseased counterparts without biochemical analysis.

 Please wait while we load your content...
Please wait while we load your content...