Mesoporous TiO2 as the support of tetraethylenepentamine for CO2 capture from simulated flue gas
Abstract
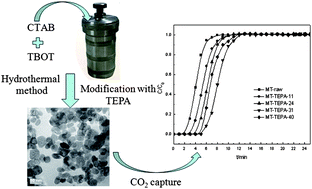
Mesoporous TiO2 (MT) was prepared by a hydrothermal method and used as the supporting material for the immobilization of tetraethylenepentamine (TEPA) to develop a new type of adsorbent for CO2 capture from flue gas. The CO2 adsorption capacity increases with the increase of the TEPA loading amounts. With the maximum TEPA loading of 31 wt% onto the MT sorbent, the maximum CO2 adsorption capacity reached 2.52 mmol of CO2 g−1 of sorbent. In the presence of an appropriate amount of water vapor, the formation of bicarbonate and hydroxylated surface on TiO2 can improve CO2 adsorption capacity from 2.64 to 2.91 mmol g−1. However, an excess of water vapor leads to a decrease in the CO2 adsorption capacity from 2.91 to 2.65 mmol g−1, probably attributed to the adsorption competition between water and CO2. The absorption/regeneration cycle was repeated and the CO2 adsorption capacity decreased from 2.52 to 2.41 mmol g−1 over 5 cycles, almost completely maintained its original CO2 adsorption capacity. In addition, the effect of long-term storage on CO2 adsorption capacity is studied. After approximately one year of storage, the CO2 capacity of the MT–TEPA-31 decreased by 28% compared with the fresh sample.

 Please wait while we load your content...
Please wait while we load your content...