Redox control in palladium catalyzed norbornene and alkyne polymerization†
Abstract
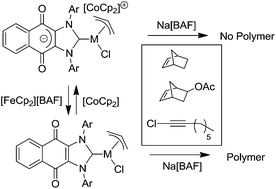
Two palladium complexes (NHC)Pd(allyl)Cl (NHC = 1,3-Ar2-naphthoquinimidazolylidene, Ar = 2,6-Me2-C6H3, 2,6-iPr2-C6H3) bearing a redox-active naphthoquinone moiety, were prepared and characterized. Electro-chemistry cyclic voltammetry and NMR studies showed that these palladium complexes could be easily reduced and re-oxidized using CoCp2 and [FeCp2][BAF] (BAF = tetrakis(3,5-bis(trifluoromethyl)phenyl)borate) reagents. The properties of these neutral and reduced palladium complexes in norbornene, 5-norbornene-2-yl acetate and 1-chloro-1-octyne polymerization were studied. The neutral complexes were active in all of these polymerization reactions in the presence of Na[BAF]. In contrast, the reduced analogues showed very low activity under the same conditions. As such, switchable polymerization could be realized in this catalytic system.
- This article is part of the themed collection: Switchable Catalysis and Related Reactions


 Please wait while we load your content...
Please wait while we load your content...