Enhancing the emission of hexa-peri-hexabenzocoronene-containing polynorbornene via electron donating, unsymmetric constitution and solvent effects†
Abstract
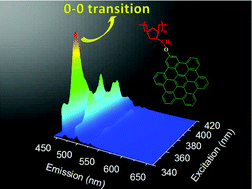
The organo-soluble and extremely stable polynorbornene PHNB-HPB with a hexaphenylbenzene (HPB) moiety was prepared by ring-opening metathesis polymerization (ROMP) followed by hydrogenation. The hexa-peri-hexabenzocoronene (HBC)-containing PHNB-HBC, which is insoluble in commercial organic solvents, was prepared by the cyclodehydrogenation of PHNB-HPB. The glass transition temperature of PHNB-HPB was 205 °C. The temperatures required for a 10% weight loss (Td10) for PHNB-HPB and PHNB-HBC were 428 °C and 456 °C under a nitrogen flow, respectively. The PHNB-HBC exhibited an ordered self-assembly in the solid state. The exfoliation of PHNB-HBC aggregates was tested by ultrasonication in various solvents. The PHNB-HBC dispersion solutions showed an intensive emission of the 0–0 transition (465 nm) due to the lower D6h symmetrical structure and alkoxy substituents. Between different solvents, the dispersion in N-cyclohexyl-2-pyrrolidone (CHP) showed the highest individual metric and the strongest 0–0 transition (465 nm), indicating higher dispersibility.


 Please wait while we load your content...
Please wait while we load your content...