Synthesis and characterization of a novel cyanostilbene derivative and its initiated polymers: aggregation-induced emission enhancement behaviors and light-emitting diode applications†
Abstract
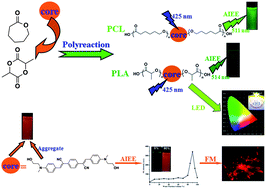
A novel cyanostilbene derivative (I) is synthesized and sufficiently characterized, which is also utilized as the initiator to prepare poly(ε-caprolactone) (I-PCL) and poly(D,L-lactide) (I-PLA) via ring-opening polymerization (ROP). Notably, molecule I exhibits superior aggregation-induced enhanced emission (AIEE) behavior in the mixed solvent of THF and water. The bulky cyano groups are linked to vinylene moieties in molecule (I) generating intramolecular coplanarity and preventing π–π stacking, which leads to enhanced emission in the aggregated state. Crystallographic data of I confirm that the existence of multiple intermolecular interactions among adjacent molecules restricts intramolecular vibration and rotation. In addition, molecule I can self-organize into ordered microtubes in appropriate THF/water mixtures exhibiting significant optical waveguide properties. Furthermore, the polymers, I-PCL and I-PLA, also show AIEE behaviors since the single chromophore I is anchored on the polymer chains. As compared with the corresponding small molecule I, the target polymers possess not only preferable thermostabilities but also the property of easy fabrication. The luminogen aggregation of the initiator is well-realized and even enlarged after polymerization, due to the linkage of polymer chains. In the end, the polymer I-PLA has a great prospect in white light-emitting diode (LED) application, which emits daylight white light.

 Please wait while we load your content...
Please wait while we load your content...