Understanding the aggregation of bis(benzoxazolyl)stilbene in PLA/PBS blends: a combined spectrofluorimetric, calorimetric and solid state NMR approach
Abstract
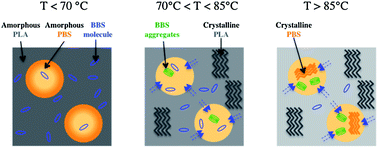
Poly(lactic acid)/poly(butylene succinate) (PLA/PBS) blends, containing a small amount of 4,4′-bis(2-benzoxazolyl)stilbene (BBS) dye, showed a chromogenic response after heating above 80 °C, due to the formation of BBS aggregates. This smart behaviour can be useful for many different applications, such as for the realization of time–temperature optical indicators, and the understanding of the processes lying behind the aggregation of chromophores is fundamental for improving their performances. In this work, the phase transitions occurring in the PLA/PBS polymeric matrix under heating were investigated in detail by combining spectrofluorimetric, DSC, and variable-temperature solid state NMR (SSNMR) measurements on both PLA- (in pellets and film) and PLA/PBS-BBS films. A detailed characterization of the phase behaviour of both PLA and PBS domains was achieved. More specifically, the SSNMR analysis of 1H FID at different temperatures was found to be a very powerful and useful approach to investigate the phase transitions of PLA, complementary to DSC. Different phase and dynamic processes, PBS and PLA cold crystallization and PLA glass transition, seem to concur, at different levels, to promote BBS aggregation. A possible mechanism for the formation of BBS aggregates under heating was proposed.

 Please wait while we load your content...
Please wait while we load your content...