Photophysical properties of rhenium(i) complexes and photosensitized generation of singlet oxygen†
Abstract
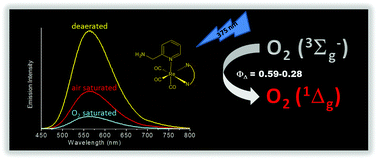
fac-[Re(ampy)(CO)3(NN)]+ complexes (ampy = 2-aminomethylpyridine and NN = 1,10-phenanthroline (phen), and 2,2′-bipyridine (bpy)) were synthesized, purified and characterized by proton nuclear magnetic resonance (1H NMR), UV-visible and Fourier-transformed infrared (FT-IR) spectroscopies, and their photophysical properties were investigated using steady state and time-resolved emission spectroscopies. The electronic absorption spectra exhibit two main absorption bands: the higher energy band, which was assigned to intraligand transition (IL), and the lower energy band assigned to metal-to-ligand charge transfer (MLCT). Both complexes showed emission at room temperature in a CH3CN solution (λmax = 560 nm, ϕ = 0.091, τ = 560 ns for fac-[Re(ampy)(CO)3(phen)]+; λmax = 568 nm, ϕ = 0.024, τ = 100 ns for fac-[Re(ampy)(CO)3(bpy)]+) and rigid media (λmax = 530 nm, τ = 3300 ns for fac-[Re(ampy)(CO)3(phen)]+; λmax = 530 nm, τ = 853 ns for fac-[Re(ampy)(CO)3(bpy)]+) arising from the lowest lying 3MLCTRe→NN excited state. Both complexes along with fac-[Re(L)(CO)3(NN)]+/0 complexes, L = Cl or pyridine, were capable of efficiently photosensitizing the generation of singlet oxygen with quantum yield in the range of 0.59–0.28. These results highlight the potential application of fac-[Re(L)(CO)3(NN)]+/0 complexes in the development of sensitizers for the generation of singlet oxygen.


 Please wait while we load your content...
Please wait while we load your content...