Small change in structure leads to large difference in protein photocleavage: two porphyrins bearing rhodanine-based pendants†
Abstract
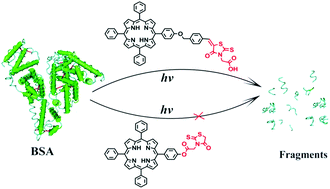
Two 5,10,15,20-tetraphenylporphyrins with one phenyl group anchored to a rhodanine-terminated side chain, RhD–TPP and RhDCOOH–TPP, were designed and synthesized, and their protein photocleavage activities were investigated using bovine serum albumin (BSA) as a model protein. Both porphyrins exhibit similar absorption spectra, fluorescence spectra, fluorescence quantum yields, and singlet oxygen (1O2) quantum yields in organic solvents due to their structure similarity. They also show similar binding affinities and binding sites toward BSA. However, RhD–TPP is nearly inactive in protein photocleavage while RhDCOOH–TPP can lead to distinct photocleavage of BSA under the same experimental conditions. Such a difference may be attributed to the different binding modes of the two porphyrin derivatives toward BSA, though the apparent binding affinities and the binding sites are similar, and consequently a great difference in the 1O2 quantum yields of the two porphyrins bound on BSA. The presence of the COOH group in RhDCOOH is proposed to play an important role, leading to less hydrophobic character and additional interactions towards BSA.

 Please wait while we load your content...
Please wait while we load your content...