Organocatalytic condensation–ring opening–annulation cascade reactions between N-Bocindolin-2-ones/benzofuran-2(3H)-ones and salicylaldehydes for synthesis of 3-arylcoumarins†
Abstract
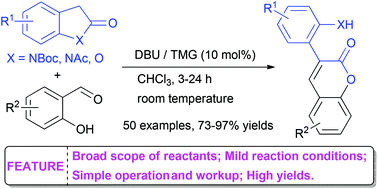
An organocatalytic cascade synthesis of 3-arylcoumarins has been developed. Mediated by 1,8-diazabicyclo[5,4,0]-undec-7-ene or tetramethylguanidine, a number of 3-arylcoumarins were obtained in good to excellent yields via condensation–ring opening–annulation between N-Bocindolin-2-ones/benzofuran-2(3H)-ones and salicylaldehydes. This method was featured by a broad scope of reactants, mild conditions, and simple operation.


 Please wait while we load your content...
Please wait while we load your content...