Syntheses and photophysical properties of BF2 complexes of curcumin analogues†
Abstract
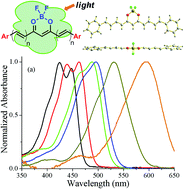
Stable tetracoordinated organoboron complexes as classic fluorescent molecules have found various applications in material and medical sciences. A new class of curcumin-BF2 complexes has been prepared from the condensation of 2,2-difluoro-1,3-dioxaborylpentadione with a variety of aldehydes and its photophysical properties were characterized. Systematic variations were observed in the absorption spectra of these curcumin-BF2 dyes in solutions, which are well correlated with their structural features, including characteristics of the aromatic groups and presence of a para electron-donating substituent. Strong fluorescence that is tunable from 500 to 800 nm via variation of the polarity of the solvent and a moderate to good fluorescence quantum yield ranging from 0.24 to 0.58 in dichloromethane were observed in curcumin-BF2 complexes 1. By contrast, their asymmetrical analogue, curcumin-BF2 complexes 3 exhibit much lower fluorescence quantum yields. Besides their excellent photostabilities, these curcumin-BF2 dyes also show singlet oxygen generating capabilities.

 Please wait while we load your content...
Please wait while we load your content...