Aromatic character and relative stability of neo-confused porphyrin tautomers and related compounds†
Abstract
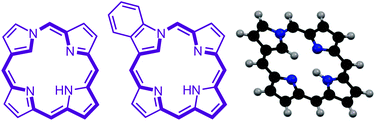
The conformations of eight neo-confused porphyrin (neo-CP) structures and seven related benzo-neo-confused porphyrins (B-neo-CPs) were minimized using DFT-B3LYP/6-311++G(d,p). In addition, in order to provide contrasts to the B-neo-CP tautomers, a series of twelve benzoporphyrin (BP) tautomers and twelve benzocarbaporphyrins (BCPs) were also analyzed, and two N-confused porphyrin (NCP) tautomers were also considered. The relative stability of the tautomers for each series was computed, and the bond lengths and bond angles were calculated. Surprisingly, all of the neo-CP and B-neo-CP tautomers were near planar. The fully conjugated members of these series showed a significant degree of bond length alternation, unlike porphyrins or the aromatic forms of NCP, BP or BCP. The most stable tautomer of neo-CP was calculated to be 27.04 kcal mol−1 higher in energy than porphyrin and 9.95 kcal mol−1 higher in energy than the most favored NCP tautomer. The most stable tautomer of B-neo-CP was similarly shown to be 26.40 kcal mol−1 higher in energy than benzoporphyrin. The favored tautomers also place the internal NH opposite to the neo-confused ring to minimize steric and lone pair–lone pair interactions, and to provide improved hydrogen bonding interactions. Nucleus independent chemical shifts (NICS) demonstrated that the fully conjugated neo-CP and B-neo-CP tautomers exhibit a significant amount of diatropic character, but these values are somewhat lower than the results obtained for porphyrins, BPs or BCPs. BCPs have three internal hydrogens, but the favored tautomer places the two pyrrolic hydrogens on each side of the indene unit to maximize hydrogen bonding interactions. Tautomers with an internal CH2 unit are also feasible, but these are energetically less favorable, have reduced diatropicity, and show significant bond length alternation. The results indicate that 18π electron delocalization pathways that pass through a fused benzene ring are less favorable than alternative delocalization pathways that bypass this unit.

 Please wait while we load your content...
Please wait while we load your content...