Direct growth of 2D nickel hydroxide nanosheets intercalated with polyoxovanadate anions as a binder-free supercapacitor electrode†
Abstract
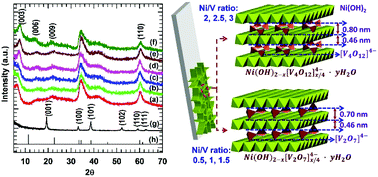
A mesoporous nanoplate network of two-dimensional (2D) layered nickel hydroxide Ni(OH)2 intercalated with polyoxovanadate anions (Ni(OH)2–POV) was built using a chemical solution deposition method. This approach will provide high flexibility for controlling the chemical composition and the pore structure of the resulting Ni(OH)2–POV nanohybrids. The layer-by-layer ordered growth of the Ni(OH)2–POV is demonstrated by powder X-ray diffraction and cross-sectional high-resolution transmission electron microscopy. The random growth of the intercalated Ni(OH)2–POV nanohybrids leads to the formation of an interconnected network morphology with a highly porous stacking structure whose porosity is controlled by changing the ratio of Ni(OH)2 and POV. The lateral size and thickness of the Ni(OH)2–POV nanoplates are ∼400 nm and from ∼5 nm to 7 nm, respectively. The obtained thin films are highly active electrochemical capacitor electrodes with a maximum specific capacity of 1440 F g−1 at a current density of 1 A g−1, and they withstand up to 2000 cycles with a capacity retention of 85%. The superior electrochemical performance of the Ni(OH)2–POV nanohybrids is attributed to the expanded mesoporous surface area and the intercalation of the POV anions. The experimental findings highlight the outstanding electrochemical functionality of the 2D Ni(OH)2–POV nanoplate network that will provide a facile route for the synthesis of low-dimensional hybrid nanomaterials for a highly active supercapacitor electrode.
- This article is part of the themed collection: Editor’s Choice: 2D Materials for Energy Storage and Conversion


 Please wait while we load your content...
Please wait while we load your content...