Natural solvent-assisted synthesis of amphiphilic co-polymeric nanomicelles for prolonged release of camptothecin delivery†
Abstract
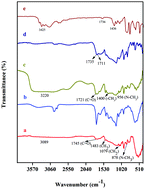
Biomaterials developed using sustainable methods and non-toxic solvents have been effectively applied as eco-friendly, sustainable reaction medium and catalysts for biological applications. This study describes the development of ε-caprolactone-citric acid nanomicelles using deep eutectic solvents (DESs) in the form of choline chloride and citric acid. The solvent formation was confirmed by FT-IR and 1H NMR spectroscopy. The highest polycondensation yield of ε-caprolactone (ε-cp) with citric acid (CA) poly(ε-cp-co-CA) was obtained in 1 : 2 DES medium. Camptothecin (CPT), used as a model anticancer agent, was encapsulated by poly(ε-cp-co-CA) micelles, which act as drug carriers. Micelle formation was determined by fluorescence analysis using pyrene (CMC = 0.5 mg mL−1). The self-assembled CPT-loaded poly(ε-cp-co-CA) micelles were characterized using FT-IR, XRD, SEM and TEM techniques. The size of the nanomicelles was observed to be ∼270 nm using dynamic light scattering (DLS) techniques. The encapsulation efficiency and in vitro profiles of CPT in poly(ε-cp-co-CA) micelles were investigated by UV spectrometry at λmax of 350 nm. The antiproliferative effect of the encapsulated CPT, which was investigated in A549 cells, suggested that ε-cp-co-CA may be a promising anticancer drug carrier.


 Please wait while we load your content...
Please wait while we load your content...