Double-shelled hollow mesoporous silica nanospheres as an acid–base bifunctional catalyst for cascade reactions†
Abstract
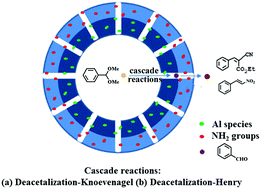
Double-shelled hollow mesoporous silica nanospheres (HMS-Al@MS-NH2) have been successfully obtained using the shell-by-shell strategy, by which the isolated acidic (–Al) and basic (–NH2) sites were spatially incorporated in different shells. The characterization results indicate that HMS-Al@MS-NH2 possesses a hollow void and mesopores in both shells, and this favors the mass transfer of the reactants and products. As a spatially isolated acid–base bifunctional catalyst, HMS-Al@MS-NH2 proved to exhibit high catalytic performances in the one-pot deacetalization-Knoevenagel cascade reaction. Under the optimized conditions, the conversion of benzaldehyde dimethyl acetal approached ca. 100% for 2 h at 110 °C, mainly attributed to the isolated acidic and basic sites and to the hollow architecture and mesopores in the shells. Notably, the catalyst could be reused up to 4 times without obvious loss of activity and selectivity, indicating the high stability of the active acidic and basic sites in the framework. Moreover, the double-shelled hollow mesoporous silica spheres are also active and selective for the other cascade sequence of the deacetalization-Henry reaction.


 Please wait while we load your content...
Please wait while we load your content...