Bis-imidazolium salts with alkyl sulfates as counterions: synthesis and liquid crystalline properties
Abstract
A series of flexibly linked bis-imidazolium salts with alkyl sulfate anions having different chain lengths (10, 12, 14) was designed and prepared starting from 1,1′-(α,ω-alkanediyl)-bisimidazole and alkyl bromides (14 or 16 carbon atoms) followed by metathesis with the corresponding sodium salts. These compounds were investigated because of their liquid crystalline properties using a combination of differential scanning calorimetry (DSC), polarized optical microscopy (POM) and temperature dependent powder X-ray diffraction (XRD). All nine bis-imidazolium compounds with alkyl sulfate anions show liquid crystalline properties which were influenced by flexible spacer length, alkyl tail length and, finally, the length of alkyl chains attached to sulfate groups. Broader mesophase ranges were found for compounds with a shorter spacer (six carbon atoms), about 70 °C. For compounds with a spacer of ten carbon atoms, higher mesophase ranges and clearing temperatures were found for longer alkyl tails (sixteen carbon atoms). Increasing the length of alkyl chains attached to sulfate anions resulted in higher clearing temperatures for compounds with the longer spacer. The variation in the carbon atom number of the alkyl sulfate anion for bis-imidazolium compounds with the shorter spacer has almost no impact on their SmA phase stability.
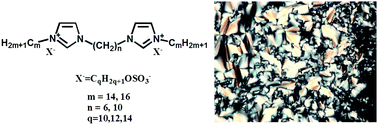


 Please wait while we load your content...
Please wait while we load your content...