Green click synthesis of β-hydroxy-1,2,3-triazoles in water in the presence of a Cu(ii)–azide catalyst: a new function for Cu(ii)–azide complexes†
Abstract
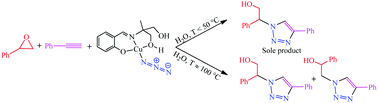
A new Cu(II)–azide complex, [Cu(H2L)(N3)]·CH3OH (1), was synthesized by the reaction of Cu(NO3)2 3H2O with 2-{[(1E)-(2-hydroxyphenyl)methylene]amino}-2-methylpropane-1,3-diol (H3L) in the presence of an azide anion and a methanol solvent. The complex was characterized by elemental analysis and spectroscopic methods (FT-IR and UV-Vis), and its structure was determined by single crystal X-ray analysis. X-ray studies showed that the asymmetric unit of this compound is a mononuclear complex of Cu(II) which converts to a dinuclear one through end-to-end (EE)-azide bridges. Complex 1 was used as the catalyst for a three-component azide–epoxide–alkyne cycloaddition reaction in water as a green solvent. The effect of the amount of catalyst, temperature and also the coordinated azide on the cycloaddition reaction was studied and the products were characterized by FT-IR, NMR and single crystal X-ray analyses. At temperatures below 50 °C just one product formed while at higher temperatures two compounds were formed by the azide attack of the epoxide ring. The structural studies indicated that complex 1 converts to a tetranuclear Cu(II) complex, [Cu4(HL)4], after catalytic reaction in the absence of sodium azide. The results showed that complex 1 is an active catalyst for preparing 1,2,3-triazole based compounds containing primary and secondary alcoholic functionalities. Based on the obtained results, a mechanism was proposed for this catalytic reaction.


 Please wait while we load your content...
Please wait while we load your content...