Facile synthesis of Pd-decorated Pt/Ru networks with highly improved activity for methanol electrooxidation in alkaline media†
Abstract
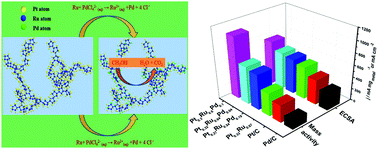
In this work, surfactant-free ternary Pt/Ru/Pd networks with tunable ratios have been prepared as anode electrocatalysts for direct methanol fuel cell reactions through a partial galvanic replacement method. Scanning electron microscopy (SEM), energy-dispersive X-ray (EDX) spectroscopy, transmission electron microscopy (TEM), X-ray diffraction (XRD) and X-ray photoelectron spectroscopy (XPS) were used to characterize the physical structure and chemical composition of the as-prepared catalysts. The electrochemical performances towards methanol electrooxidation of Pt0.3Ru0.6Pd0.1, Pt0.27Ru0.54Pd0.19, Pt0.32Ru0.64Pd0.04, Pt0.33Ru0.67, Pt/Ru, commercial Pt/C, PtRu/C and Pd/C were evaluated. The results proved that the Pt/Ru/Pd catalysts displayed much larger electrochemically active surface areas and higher current densities as well as superior long-term stability in comparison with the Pt/Ru, commercial Pd/C, PtRu/C and Pt/C catalysts. The enhanced electrocatalytic performances are primarily attributed to the synergistic effect of Pt, Ru and Pd together with the 3D nanostructure. The achievements acquired by this facile synthetic pathway and the prepared catalytic materials in this contribution are beneficial for opening a novel way in tuning 3D multimetallic nanostructures for boosting the development of practical commercialization applications of electrocatalysts for methanol electrooxidation.


 Please wait while we load your content...
Please wait while we load your content...