Organic solvent soluble methyltriphenylphosphonium peroxodisulfate: a novel oxidant for the synthesis of polyaniline and the thus prepared polyaniline in high performance supercapacitors
Abstract
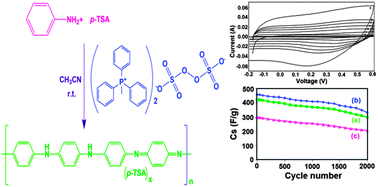
Polyaniline samples are prepared via a chemical polymerization pathway of aniline in water medium. The aim of this work is to carry out the polymerization of aniline in an organic solvent medium. Since the oxidant, methyltriphenylphosphonium peroxodisulfate (MTPPS), is soluble in acetonitrile solvent, aniline was oxidatively polymerized in the presence of p-toluenesulfonic acid and MTPPS via a solution polymerization pathway (PANI-Sol). For comparison, a polyaniline salt was synthesized via emulsion (PANI-Eml) and interfacial (PANI-Int) polymerization pathways. These three polymerization pathways lead to the formation of polyaniline-p-toluenesulfonate salt (PANI-p-TSA) in acetonitrile solvent as a green approach. The powder form of PANI-p-TSA was isolated from the solution by precipitation with acetone solvent. Formation of polyaniline was confirmed by infrared and X-ray diffraction spectral results. PANI-Sol, PANI-Eml, and PANI-Int showed nanocrystal, nanopetal, and nanospherical morphology, respectively. The yield and the conductivity of the polyaniline salt prepared in the present study are comparable with those of the conventional polyaniline salt. The specific capacitances of the PANI-Eml, PANI-Sol, and PANI-Int were 460, 424 and 300 F g−1, respectively, which was higher than the conventional PANI-p-TSA salt. The energy densities were 23, 21.2, and 15 W h kg−1, respectively at a power density of 200 W kg−1. After 2000 charge–discharge cycles, the retention in the specific capacitance values of the polyaniline salts were 70.4% (PANI-Sol), 73% (PANI-Eml) and 69.3% (PANI-Int).

 Please wait while we load your content...
Please wait while we load your content...