Cycloalkenyl nonaflates as electrophilic cross-coupling substrates for palladium catalyzed C–N bond forming reactions with enolizable heterocycles under microwave enhanced conditions†
Abstract
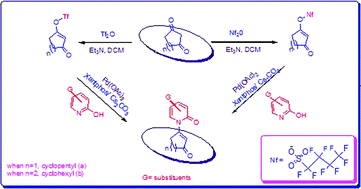
Microwave-mediated, palladium catalyzed C–N bond forming reactions with activated cycloalkenyl nonaflates and enolizable heterocycles using cesium carbonate as a base and a catalytic system employing Pd(OAc)2/Xantphos or Pd(OAc)2/dppp were found to be effective in driving the reactions to completion. This method resulted in good to excellent yields of the coupled products (22 examples, 25–90%) in short reaction time (30–60 min). Under optimum conditions, cycloalkenyl nonaflates were found to be an effective alternative to analogous triflates in C–N bond forming processes due to their increased stability under the reaction conditions. The use of tetrabutylammonium bromide (Bu4NBr) as an additive in these transformations proved to be effective and resulted in better yields of the coupled products.

 Please wait while we load your content...
Please wait while we load your content...