Removal of uranium(vi) from aqueous solutions by surface modified magnetic Fe3O4 particles
Abstract
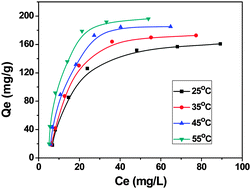
We prepared surface modified magnetic Fe3O4 particles (Fe3O4@APTMS) which could efficiently remove uranium(VI) ions from aqueous solutions. Batch experiments have been conducted to study the effects of initial pH, adsorbent dosage, shaking time and temperature on uranium sorption efficiency. It is found that the maximum adsorption capacity of Fe3O4@APTMS toward uranium(VI) is 151.80 mg g−1, displaying a high efficiency for the removal of uranium(VI) ions. The metal-loaded Fe3O4@APTMS particles could be recovered readily from aqueous solution by magnetic separation and regenerated easily by alkali treatment. The present study suggests that magnetic Fe3O4@APTMS composite particles can be used as an effective and recyclable adsorbent for the removal of uranium(VI) from aqueous solutions.

 Please wait while we load your content...
Please wait while we load your content...