Structural characteristics that make chlorophylls green: interplay of hydrocarbon skeleton and substituents
Abstract
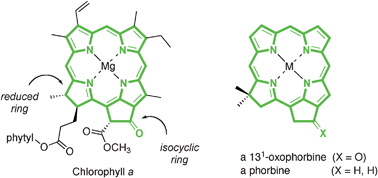
Understanding the effects of substituents on natural photosynthetic pigments is essential for gaining a deep understanding of why such pigments were selected over the course of evolution for use in photosynthetic systems. This knowledge should provide for a more thoughtful design of artificial light-harvesting systems. The hydrocarbon skeleton of all

 Please wait while we load your content...
Please wait while we load your content...