Synthesis and anti-tubercular activity of conformationally-constrained and bisquinoline analogs of TMC207†
Abstract
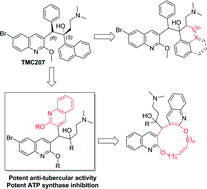
One of the most significant breakthroughs in the battle against tuberculosis is the recent approval of the quinoline compound, TMC207, for the treatment of drug-resistant tuberculosis. To gain insight into the molecular determinants of the activity of TMC207 and to evaluate the scope of quinoline compounds as anti-tubercular agents, we synthesized a series of TMC207 derivatives and evaluated their anti-tubercular activity. Making the lateral chain of the drug rigid by linking it to an adjacent phenyl substituent resulted in a decrease in activity. In contrast, replacing a phenyl substituent of TMC207 with a quinoline moiety gave bisquinolines that demonstrated potent anti-tubercular activity in in vitro experiments, in ex vivo mouse bone marrow macrophage assays, and also in the in vivo mouse model of the disease. These results provide new guiding principles for modifying the TMC207 scaffold to develop efficacious anti-tubercular drugs and set the stage for the development of bisquinolines as a promising new class of anti-tubercular agents.

 Please wait while we load your content...
Please wait while we load your content...