Activation mechanism of claudin-4 by ephrin type-A receptor 2: a molecular dynamics approach†
Abstract
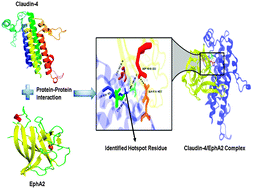
Claudin-4 is a crucial component in the tight junction which is involved in the organization of a cellular barrier. Claudin-4 is found to be up-regulated in various malignancies and is activated by forming a complex with an ephrin A2 receptor. In this computational study, we propose a rational model for the claudin-4–ephrinA2 complex which is in agreement with the experimental result. The complex model has been obtained through protein–protein docking, interface residue scanning, in silico alanine mutations and extensive molecular dynamics simulations. The docking model envisages the important residues present in the first extracellular loop of claudin-4 that plays an active role in protein–protein interaction and stability. A 30 nanosecond molecular dynamics simulation of the complex revealed a higher stability by which the number of hydrogen bond interactions increased in the complex interface. Both the molecular dynamics simulations and in silico alanine mutations revealed the involvement of Lys65 (claudin-4) as one of the prime residues in the complex interface that is actively engaged in the binding mechanism with its counterpart. We postulate that the novel hotspot, Lys65 of claudin-4 can be targeted through structure based inhibitor design, which could alter the effect of the claudin-4–ephrinA2 binding mechanism in aggressive metastatic tumors.

 Please wait while we load your content...
Please wait while we load your content...